3-dehydroquinate dehydratase
| 3-dehydroquinate dehydratase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
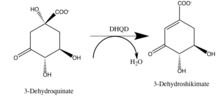 The third step of the shikimate pathway is catalyzed by DHQD | |||||||||
| Identifiers | |||||||||
| EC number | 4.2.1.10 | ||||||||
| CAS number | 9012-66-2 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / EGO | ||||||||
| |||||||||
| Type I 3-dehydroquinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
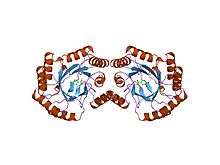 The structure of type i 3-dehydroquinate dehydratase from salmonella typhi | |||||||||
| Identifiers | |||||||||
| Symbol | DHquinase_I | ||||||||
| Pfam | PF01487 | ||||||||
| Pfam clan | CL0036 | ||||||||
| InterPro | IPR001381 | ||||||||
| PROSITE | PDOC00789 | ||||||||
| SCOP | 2dhq | ||||||||
| SUPERFAMILY | 2dhq | ||||||||
| CDD | cd00502 | ||||||||
| |||||||||
| Dehydroquinase class II | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | DHquinase_II | ||||||||
| Pfam | PF01220 | ||||||||
| PROSITE | PDOC00789 | ||||||||
| SCOP | 2dhq | ||||||||
| SUPERFAMILY | 2dhq | ||||||||
| CDD | cd00466 | ||||||||
| |||||||||
In enzymology, a 3-dehydroquinate dehydratase (EC 4.2.1.10) is an enzyme that catalyzes the chemical reaction
- 3-dehydroquinate 3-dehydroshikimate + H2O
Hence, this enzyme has one substrate, 3-dehydroquinate, and two products, 3-dehydroshikimate and H2O.
This enzyme belongs to the family of lyases, specifically the hydro-lyases, which cleave carbon-oxygen bonds. This enzyme participates in phenylalanine, tyrosine and tryptophan biosynthesis.
Discovery
The shikimate pathway was determined to be a major biosynthetic route for the production of aromatic amino acids through the research of Bernhard Davis and David Sprinson.[1]
Role in the shikimate pathway
3-Dehydroquinate Dehydratase is an enzyme that catalyzes the third step of the shikimate pathway. The shikimate pathway is a biosynthetic pathway that allows plants, fungi, and bacteria to produce aromatic amino acids.[2] Mammals do not have this pathway, meaning that they must obtain these essential amino acids through their diet. Aromatic Amino acids include Phenylalanine, Tyrosine, and Tryptophan.[1]
This enzyme dehydrates 3-Dehydroquinate, converting it to 3-Dehydroshikimate, as indicated in the diagram on the right. This is the third step in the Shikimate pathway. It belongs to the family of lyases, specifically the hydro-lyases, which cleave carbon-oxygen bonds. The systematic name of this enzyme class is 3-dehydroquinate hydro-lyase (3-dehydroshikimate-forming). This enzyme is one of the few examples of convergent evolution. The two separate versions of this enzyme have different amino acid sequences.[2]
3-Dehydroquinate dehydratase is also commonly referred to as Dehydroquinate dehydratase and DHQD. Other names include 3-dehydroquinate hydrolase, DHQase, 3-dehydroquinase, 5-dehydroquinase, dehydroquinase, 5-dehydroquinate dehydratase, 5-dehydroquinate hydro-lyase, and 3-dehydroquinate hydro-lyase.[2]
Evolutionary origins
Purposes of the products
The aromatic amino acids produced by the shikimate acid pathway are used by higher plants as protein building blocks and as precursors for several secondary metabolites. Examples of such secondary metabolites are plant pigments and compounds to defend against herbivores, insects, and UV light. The specific aromatic secondary metabolites produced, as well as when and in what quantities they are produced in, varies across different types of plants. Mammals consume essential amino acids in their diets, converting them to precursors for important substances such as neurotransmitters.
Convergent evolution
As mentioned previously, two classes of 3-Dehydroquinate Dehydratase exist, known as types I and II. These two versions have different amino acid sequences and different secondary structures. Type I is present in fungi, plants, and some bacteria, for the biosynthesis of chorismate. It catalyzes the cis-dehydration of 3-Dehydroquinate via a covalent imine intermediate. Type I is heat liable and has Km values in the low micromolar range. Type II is present in the quinate pathway of fungi and the shikimate pathway of most bacteria. It catalyzes a trans-dehydration using an enolate intermediate. It is heat stable and has Km values one or two orders of magnitude higher than the Type I Km values.[1]
The best studied type I enzyme is from Escherichia coli (gene aroD) and related bacteria. It is a homodimeric protein. In fungi, dehydroquinase is part of a multifunctional enzyme which catalyses five consecutive steps in the shikimate pathway. A histidine is involved in the catalytic mechanism.[3]
Other purposes
3-Dehydroquinate Dehydratase is also an enzyme present in the process of the degradation of quinate. Both 3-Dehydroquinate and 3-Dehydroshikimate are intermediates in the reaction mechanism. The following image shows this process in Quinate Degradation.[1]
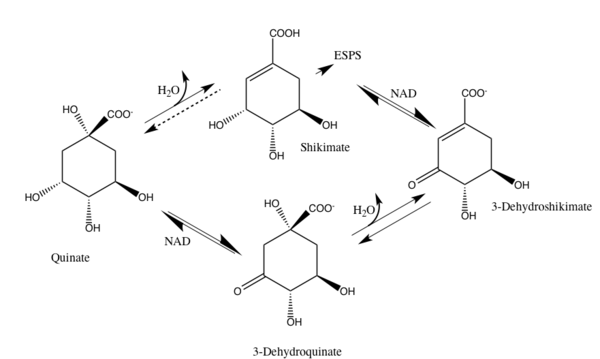
Structure

Applications
The Shikimate pathway has become a focus of research into the development of herbicides and antimicrobial agents because it is an essential pathway in many plants, bacteria, and parasites but does not exist in mammals.[1]
Inhibitors of the shikimate pathway in mycobacterium have the potential of treating tuberculosis.[4][5]
Most of the 3-dehydroquinate-dehydratase in bacteria and higher plants is type I DHQD.[1]
References
- 1 2 3 4 5 6 Herrmann KM (July 1995). "The Shikimate Pathway: Early Steps in the Biosynthesis of Aromatic Compounds". The Plant Cell. 7 (7): 907–919. doi:10.1105/tpc.7.7.907. PMC 160886
 . PMID 12242393.
. PMID 12242393. - 1 2 3 Herrmann KM (January 1995). "The shikimate pathway as an entry to aromatic secondary metabolism". Plant Physiology. 107 (1): 7–12. doi:10.1104/pp.107.1.7. PMC 161158
 . PMID 7870841.
. PMID 7870841. - ↑ Deka RK, Kleanthous C, Coggins JR (November 1992). "Identification of the essential histidine residue at the active site of Escherichia coli dehydroquinase". The Journal of Biological Chemistry. 267 (31): 22237–42. PMID 1429576.
- ↑ Dias MV, Snee WC, Bromfield KM, Payne RJ, Palaninathan SK, Ciulli A, Howard NI, Abell C, Sacchettini JC, Blundell TL (June 2011). "Structural investigation of inhibitor designs targeting 3-dehydroquinate dehydratase from the shikimate pathway of Mycobacterium tuberculosis". The Biochemical Journal. 436 (3): 729–39. doi:10.1042/BJ20110002. PMID 21410435.
- ↑ Reichau S, Jiao W, Walker SR, Hutton RD, Baker EN, Parker EJ (May 2011). "Potent inhibitors of a shikimate pathway enzyme from Mycobacterium tuberculosis: combining mechanism- and modeling-based design". The Journal of Biological Chemistry. 286 (18): 16197–207. doi:10.1074/jbc.M110.211649. PMC 3093739
 . PMID 21454647.
. PMID 21454647.
Further reading
- Mitsuhashi S, Davis BD (September 1954). "Aromatic biosynthesis. XII. Conversion of 5-dehydroquinic acid to 5-dehydroshikimic acid dy 5-dehydroquinase". Biochimica et Biophysica Acta. 15 (1): 54–61. doi:10.1016/0006-3002(54)90093-1. PMID 13198937.
- Mitsuhashi S, Davis BD (October 1954). "Aromatic biosynthesis. XIII. Conversion of quinic acid to 5-dehydroquinic acid by quinic dehydrogenase". Biochimica et Biophysica Acta. 15 (2): 268–80. doi:10.1016/0006-3002(54)90069-4. PMID 13208693.
This article incorporates text from the public domain Pfam and InterPro IPR001381