Acetylcarnosine
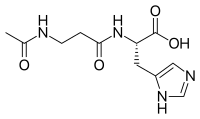 | |
| Names | |
|---|---|
| IUPAC name
(S)-2-(3-Acetamidopropanoylamino)-3-(1H-imidazol-5-yl)propanoic acid | |
| Other names
N-(N-Acetyl-β-alanyl)histidine; N-Acetyl-L-carnosine; N-Acetylcarnosine | |
| Identifiers | |
| 56353-15-2 | |
| 3D model (Jmol) | Interactive image |
| Abbreviations | NAC |
| ChemSpider | 8079136 |
| ECHA InfoCard | 100.054.640 |
| PubChem | 9903482 |
| |
| |
| Properties | |
| C11H16N4O4 | |
| Molar mass | 268.27 g/mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
N-Acetylcarnosine (NAC) is a naturally occurring[1] compound chemically related to the dipeptide carnosine. The NAC molecular structure is identical to carnosine with the exception that it carries an additional acetyl group. The acetylation makes NAC more resistant to degradation by carnosinase, an enzyme that breaks down carnosine to its constituent amino acids, beta-alanine and histidine.[2]
Actions
Carnosine and metabolic derivatives of carnosine, including NAC, are found in a variety of tissues but particularly muscle tissue.[1] These compounds have varying degrees of activity as free radical scavengers.[1] NAC is particularly active against lipid peroxidation in the different parts of the lens in the eye.[3] It is the active ingredient of eye drops used in order to prevent or treat cataracts.
Research
During early experiments performed at the Moscow Helmholtz Research Institute for Eye Diseases, it was shown that NAC (1% concentration), was able to pass from the cornea to the aqueous humour after about 15 to 30 minutes.[4]
It is believed that NAC is deacetylated (loses its acetyl group) and transforms into carnosine, which then acts as an antioxidant and against glycation.[5] In another study NAC was reported as effective in improving vision in cataract patients and reduced the appearance of cataract.[6] The authors called this ‘a snow melting effect’ referring to the slow reduction of the cataractous tissues in the lens following the use of NAC eye drops. Transparency of the lens improved after using NAC eye drops at a concentration of 1% twice a day for four months. These results were relevant to all forms of cataract, mild or severe, although other studies found the most beneficial effect was in relation to early forms of cataract.[7] This was a landmark study which caused many commercial operators to manufacture their own brands of anti-cataract NAC eye drops.[8]
In a 2004 trial of 90 canine eyes with cataracts, led by researcher Mark Babizhayev, NAC was reported to have performed better than placebo in positively affecting lens clarity.[9] The Babizhayev group later published a placebo-controlled clinical trial of NAC in 76 human eyes with mild to advanced cataracts and reported similar positive results for NAC. However, a 2007 scientific review of the current literature discussed the limitations of the clinical trial, noting that the study had low statistical power, a high dropout rate and "insufficient baseline measurement to compare the effect of NAC," concluding that "a separate larger trial is needed to justify the benefit of long-term NAC therapy."[10]
The Royal College of Ophthalmologists issued the following public statement about NAC as of August 2008:
The evidence for the effectiveness of N-acetyl carnosine eye drops is based on experience on a small number of cases carried out by a Russian researcher team [Babizhayev]. To date, the research has not been corroborated and the results replicated by others. The long-term effect is unknown. Unfortunately, the evidence to date does not support the 'promising potential' of this drug in cataract reversal. More robust data from well conducted clinical trials on adequate sample sizes will be required to support these claims of efficacy. Furthermore, we do not feel the evidence base for the safety is in any way sufficient to recommend its use in the short term. More research is needed.[11]
Babizhayev and colleagues, of the US-based company Innovative Vision Products (IVP), which markets NAC treatments, published another human clinical trial in 2009. They reported positive results for NAC as well as arguing "only certain formulas designed by IVP..are efficacious in the prevention and treatment of senile cataract for long-term use."[12]
In a 2010 book on ocular disease, the current state of this subject is summarized as follows:
Carnosine (β-alanyl-L-histidine), and its topical prodrug formulation N-acetylcarnosine (NAC), is advertised (especially on the internet) to treat a range of ophthalmic disorders associated with oxidative stress, including age-related and diabetic cataracts. No convincing animal studies or masked clinical trials have been reported.[13]
References
- 1 2 3 Boldyrev A, Abe H (February 1999). "Metabolic transformation of neuropeptide carnosine modifies its biological activity" (PDF). Cell. Mol. Neurobiol. 19 (1): 163–75. doi:10.1023/a:1006914028195. PMID 10079975.
- ↑ Pegova A, Abe H, Boldyrev A (December 2000). "Hydrolysis of carnosine and related compounds by mammalian carnosinases". Comp. Biochem. Physiol. B, Biochem. Mol. Biol. 127 (4): 443–6. doi:10.1016/S0305-0491(00)00279-0. PMID 11281261.
- ↑ Bonnefont-Rousselot D (2001). "Antioxidant and anti-AGE therapeutics". J Soc Biol. 195 (4): 391–398. PMID 11938556.
- ↑ Babizhayev MA, Yermakova VN, Sakina NL, et al. (1996). "N alpha acetylcarnosine as a pro-drug of L-carnosine in ophthalmic application as antioxidant". Clin Chim Acta. 254 (1-2): 199–121. doi:10.1016/0009-8981(96)06356-5. PMID 8894306.
- ↑ Boldyrev AA, Dupin AM, Bunin AY, et al. (1987). "The antioxidative properties of carnosine, a natural histidine-containing dipeptide". Biochem Int. 15 (6): 1105–1113. PMID 3326603.
- ↑ Babizhayev MA, Deyev AI, Yermakova VN, et al. (2002). "Efficacy of N-acetylcarnosine in the treatment of cataracts". Drugs R D. 3 (2): 87–103. doi:10.2165/00126839-200203020-00004. PMID 12001824.
- ↑ Williams DL, Munday P (2006). "The effect of a topical antioxidant formulation including N-acetyl carnosine on canine cataract: a preliminary study". Vet Ophthalmol. 9 (5): 311–6. doi:10.1111/j.1463-5224.2006.00492.x. PMID 16939459.
- ↑ Marios Kyriazis. The Cataract Cure. iUniverse, New York 2005
- ↑ Babizhayev MA, Deyev AI, Yermakova VN, Brikman IV, Bours J (2004). "Lipid peroxidation and cataracts: N-acetylcarnosine as a therapeutic tool to manage age-related cataracts in human and in canine eyes". Drugs R D. 5 (3): 125–39. doi:10.2165/00126839-200405030-00001. PMID 15139774.
- ↑ Toh T, Morton J, Coxon J, Elder MJ (2007). "Medical treatment of cataract". Clin. Experiment. Ophthalmol. 35 (7): 664–71. doi:10.1111/j.1442-9071.2007.01559.x. PMID 17894689.
- ↑ "N-Acetyl Carnosine for cataracts". Retrieved July 15, 2012.
"Clinical Guidelines". Retrieved August 16, 2012. - ↑ Babizhayev MA, Burke L, Micans P, Richer SP (2009). "N-Acetylcarnosine sustained drug delivery eye drops to control the signs of ageless vision: glare sensitivity, cataract amelioration and quality of vision currently available treatment for the challenging 50,000-patient population". Clin Interv Aging. 4: 31–50. doi:10.2147/cia.s4090. PMC 2685223
 . PMID 19503764.
. PMID 19503764. - ↑ Levin, Leonard A; Albert DM (2010). Ocular Disease: Mechanisms and Management: Expert Consult - Online and Print, 1e. Saunders. p. 249. ISBN 0702029831.