Acyl-CoA
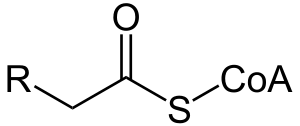
Acyl-CoA is a group of coenzymes involved in the metabolism of fatty acids. It is a temporary compound formed when coenzyme A (CoA) attaches to the end of a long-chain fatty acid inside living cells. The compound undergoes beta oxidation, forming one or more molecules of acetyl-CoA. This, in turn, enters the citric acid cycle, eventually forming several molecules of ATP.
Functions
Fatty acid activation
To be oxidatively degraded, a fatty acid must first be activated in a two-step reaction catalyzed by acyl-CoA synthetase. First, the fatty acid displaces the diphosphate group of ATP, then coenzyme A (HSCoA) displaces the AMP group to form an acyl-CoA. The acyladenylate product of the first step has a large free energy of hydrolysis and conserves the free energy of the cleaved phosphoanhydride bond in ATP. The second step, transfer of the acyl group to CoA (the same molecule that carries acetyl groups as acetyl-CoA), conserves free energy in the formation of a thioester bond. Consequently, the overall reaction has a free energy change near zero:
- Fatty acid + CoA + ATP ⇌ Acyl-CoA + AMP + PPi
Subsequent hydrolysis of the product PPi (by the enzyme inorganic pyrophosphatase) is highly exergonic, and this reaction makes the formation of acyl-CoA spontaneous and irreversible.
Fatty acids are activated in the cytosol, but oxidation occurs in the mitochondria. Because there is no transport protein for CoA adducts, acyl groups must enter the mitochondria via a shuttle system involving the small molecule carnitine.[1]
See also
References
- ↑ Pratt C.W., Cornely, K. Essential Biochemistry. John Wiley & Sons, Inc. (2004)
External links
- Acyl Coenzyme A at the US National Library of Medicine Medical Subject Headings (MeSH)
| ||||||||||||||||||||