Akabori amino-acid reaction
There are several Akabori amino acid reactions, which are named after Shiro Akabori (1900–1992), a Japanese chemist.
In the first reaction, an α-amino acid is oxidised by heating it with an oxidizing sugar.[1][2] This reaction has been used to synthesize dichlorophthalimido derivatives for the analysis of peptides, since the mass spectra of those derivatives are easily recognized.[3]
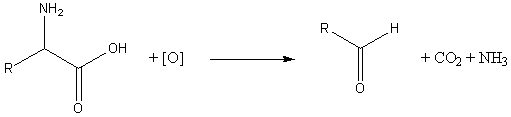
In the second reaction, an α-amino acids and esters are reduced by sodium amalgam and ethanolic HCl to give α-amino aldehydes.[4][5] This process is conceptually similar to the Bouveault–Blanc reduction except that is pauses at the aldehyde stage.
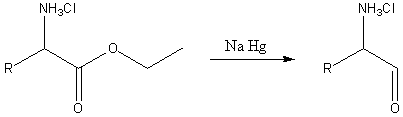
See also
References
- ↑ S. Akabori (1931). "Studies on amino acids and its derivatives. I. Oxidation of sugars for a-amino acids". J. Chetn. Soc. Japan, Pure Chem. Sect. 52: 606–810.
- ↑ S. Akabori (1933). "Oxydativer Abbau von α-Amino-säuren durch Zucker1)". Chemische Berichte. 66 (2): 143. doi:10.1002/cber.19330660213.
- ↑ "Akabori Amino Acid Reaction". Comprehensive Organic Name Reactions and Reagents. 8: 29–32. 2010. doi:10.1002/9780470638859.conrr008.
- ↑ A. Lawson, H.V. Motley (1955). "2-Mercaptoglyoxalines. Part IX. The preparation of 1 : 5-disubstituted 2-mercaptoglyoxalines from α-amino-acids". J. Chem. Soc.: 1695. doi:10.1039/jr9550001695.
- ↑ A. Lawson (1956). "63. The reaction of cyanamide with α-amino-acetals and α-amino-aldehydes". J. Chem. Soc.: 307. doi:10.1039/jr9560000307.
This article is issued from Wikipedia - version of the 7/29/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.