Copper(I) bromide
-bromide-sample.jpg) | |
 | |
| Names | |
|---|---|
| Other names
Cuprous bromide | |
| Identifiers | |
| 7787-70-4 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 22995 |
| ECHA InfoCard | 100.029.210 |
| PubChem | 24593 |
| |
| |
| Properties | |
| CuBr | |
| Molar mass | 143.45 g/mol |
| Appearance | green powder (see text) |
| Density | 4.71 g/cm3, solid |
| Melting point | 492 °C (918 °F; 765 K) |
| Boiling point | 1,345 °C (2,453 °F; 1,618 K) |
| slightly soluble | |
| Solubility | soluble in HCl, HBr, ammonium hydroxide, acetonitrile negligible in acetone, sulfuric acid |
| Refractive index (nD) |
2.116 |
| 1.46 D | |
| Hazards | |
| Flash point | Non-flammable |
| US health exposure limits (NIOSH): | |
| PEL (Permissible) |
TWA 1 mg/m3 (as Cu)[1] |
| REL (Recommended) |
TWA 1 mg/m3 (as Cu)[1] |
| IDLH (Immediate danger) |
TWA 100 mg/m3 (as Cu)[1] |
| Related compounds | |
| Other anions |
Copper(I) chloride Copper(I) iodide |
| Other cations |
Silver(I) bromide Copper(II) bromide Mercury(I) bromide |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
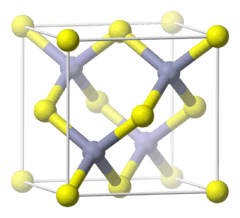
Copper(I) bromide is the chemical compound with the formula CuBr. This diamagnetic solid adopts a polymeric structure akin to that for zinc sulfide. The compound is widely used in the synthesis of organic compounds.
Preparation, basic properties, structure
The pure solid is colourless, although samples are often colored due to the presence of copper(II) impurities (see picture).[2] The copper(I) ion also oxidizes easily in air. It is commonly prepared by the reduction of cupric salts with sulfite in the presence of bromide.[3] For example, the reduction of copper(II) bromide with sulfite yields copper(I) bromide and hydrogen bromide:
- 2 CuBr2 + H2O + SO32− → 2 CuBr + SO42− + 2 HBr
CuBr is insoluble in most solvents due to its polymeric structure, which features four-coordinated, tetrahedral Cu centers interconnected by bromide ligands (ZnS structure). Upon treatment with Lewis bases, CuBr converts to molecular adducts. For example with dimethyl sulfide, the colorless complex is formed:[4]
- CuBr + S(CH3)2 → CuBr(S(CH3)2)
In this coordination complex, the copper is two-coordinate, with a linear geometry. Other soft ligands afford related complexes. For example triphenylphosphine gives CuBr(P(C6H5)3), although this species has a more complex structure. Thermal excitation of copper(I) bromide vapour yields a blue violet emission which is of greater saturation than known copper(I) chloride emission.[5] Copper(I) bromide is hence an advantageous emitter in pyrotechnic flames.
Applications in organic chemistry
In the Sandmeyer reaction, CuBr is employed to convert diazonium salts into the corresponding aryl bromides:[3]
- ArN2+ + CuBr → ArBr + N2 + Cu+
The aforementioned complex CuBr(S(CH3)2) is widely used to generate organocopper reagents.[4] Related CuBr complexes are catalysts for Atom Transfer Radical Polymerization and Cu-catalyzed Cross-Dehydrogenative Couplings (CDC).
References
- 1 2 3 "NIOSH Pocket Guide to Chemical Hazards #0150". National Institute for Occupational Safety and Health (NIOSH).
- ↑ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- 1 2 This report gives a procedure for generating CuBr: Jonathan L. Hartwell (1955). "o-Chlorobromobenzene". Org. Synth.; Coll. Vol., 3, p. 185.
- 1 2 Jarowicki, K.; Kocienski, P. J.; Qun, L. "1,2-Metallate Rearrangement: (Z)-4-(2-Propenyl)-3-Octen-1-ol" Organic Syntheses, Collected Volume 10, p.662 (2004).http://www.orgsyn.org/orgsyn/pdfs/V79P0011.pdf
- ↑ E.-C. Koch, Spectral Investigation and Color Properties of Copper(I) Halides CuX (X=F, Cl, Br, I) in Pyrotechnic Combustion Flames, Propellants Explos. Pyrotech. 2015, 40, 798-802.
External links
| Wikimedia Commons has media related to Copper(I) bromide. |