Antimony tetroxide
 | |
| Names | |
|---|---|
| IUPAC name
antimony(III,V) oxide | |
| Identifiers | |
| 1332-81-6 | |
| ECHA InfoCard | 100.032.554 |
| PubChem | 74002 |
| Properties | |
| SbO2; Sb2O4 | |
| Molar mass | 153.7588; 307.5176 g/mol |
| Appearance | white solid |
| Density | 6.64 g/cm3 (orthorhombic form) [1] |
| Melting point | > 930 °C (1,710 °F; 1,200 K) (decomposes) |
| Boiling point | decomposes |
| insoluble | |
| Refractive index (nD) |
2.0 |
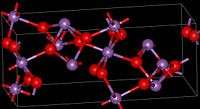
| Structure | |
| orthorhombic | |
| Hazards | |
| NFPA 704 | |
| US health exposure limits (NIOSH): | |
| PEL (Permissible) |
TWA 0.5 mg/m3 (as Sb)[2] |
| REL (Recommended) |
TWA 0.5 mg/m3 (as Sb)[2] |
| Related compounds | |
| Related compounds |
Antimony trioxide Antimony pentoxide |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Antimony tetroxide is an inorganic compound with the formula Sb2O4. This material, which exists as the mineral cervantite,[3] is white but reversibly yellows upon heating. The material, with empirical formula SbO2, is called antimony tetroxide to signify the presence of two kinds of Sb centers.
Formation and structure
The material forms when Sb2O3 is heated in air:[4]
- Sb2O3 + 0.5 O2 → Sb2O4 ΔH = −187 kJ/mol
At 800 °C, antimony(V) oxide loses oxygen to give the same material:
- Sb2O5 → Sb2O4 + 0.5 O2 ΔH = −64 kJ/mol
The material is mixed valence, containing both Sb(V) and Sb(III) centers. Two polymorphs are known, one orthorhombic (shown in the infobox) and one monoclinic.[1] Both forms feature octahedral Sb(V) centers arranged in sheets with distorted Sb(III) centers bound to four oxides.
References
- 1 2 Amador, J.; Puebla, E. Gutierrez; Monge, M. A.; Rasines, I.; Valero, C. Ruiz (1988). "Diantimony Tetraoxides Revisited". Inorganic Chemistry. 27: 1367–1370. doi:10.1021/ic00281a011.
- 1 2 "NIOSH Pocket Guide to Chemical Hazards #0036". National Institute for Occupational Safety and Health (NIOSH).
- ↑ "Cervantite". Webminerals. Retrieved 2009-06-06.
- ↑ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
This article is issued from Wikipedia - version of the 10/18/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.
