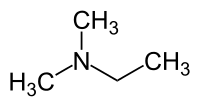
N,N-Dimethylethylamine
 | |
| Names | |
|---|---|
| Preferred IUPAC name
N,N-Dimethylethan-1-amine | |
| Other names
Ethyl(dimethyl)amine | |
| Identifiers | |
| 598-56-1 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 11230 |
| ECHA InfoCard | 100.009.038 |
| 5523 | |
| PubChem | 11723 |
| |
| Properties | |
| C4H11N | |
| Molar mass | 73.14 g·mol−1 |
| Appearance | Volatile liquid at room temp. |
| Density | 0.7±0.1 g/cm3 |
| Melting point | −140 °C (−220 °F; 133 K) |
| Boiling point | 36.5 °C (97.7 °F; 309.6 K) |
| Vapor pressure | 495.4±0.1 mmHg |
| Acidity (pKa) | 10.16 (for the conjugate acid) (H2O)[1] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
N,N-Dimethylethylamine (DMEA), sometimes referred to as dimethylethylamine, is an organic compound with formula C4H11N. It is an industrial chemical that is mainly used in foundries as a catalyst for sand core production.[2] Dimethylethylamine is a malodorous, volatile liquid at room temperature that is excreted at greater concentrations with larger dietary intake of trimethylamine.[1] It has recently been identified as an agonist of human trace amine-associated receptor 5.[3]
See also
References
- 1 2 "N,N-Dimethylethylamine". Toxnet. Hazardous Substance Data Bank. Retrieved 4 May 2014.
The aim was to study the effect of trimethylamine (TMA) on the metabolism of the industrial catalyst N,N-dimethylethylamine to ascertain whether biological monitoring of industrial exposure to N,N-dimethylethylamine is compromised and excretion of the malodorous N,N-dimethylethylamine in sweat and urine is increased by dietary intake of TMA....Although the increased urinary and hidrotic excretion of N,N-dimethylethylamine may contribute to body odor problems, they were primarily due to TMA excretion, which is much the greater.
- ↑ "Dimethylethylamine". BASF The Chemical Company. Retrieved 4 May 2014.
- ↑ Wallrabenstein I, Kuklan J, Weber L, Zborala S, Werner M, Altmüller J, Becker C, Schmidt A, Hatt H, Hummel T, Gisselmann G (2013). "Human trace amine-associated receptor TAAR5 can be activated by trimethylamine". PLoS ONE. 8 (2): e54950. doi:10.1371/journal.pone.0054950. PMC 3564852
 . PMID 23393561.
. PMID 23393561.
This article is issued from Wikipedia - version of the 9/5/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.