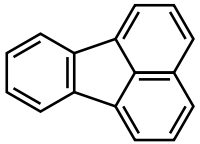
Fluoranthene
 | |
| | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Fluoranthene | |
| Other names
Benzo[jk]fluorene | |
| Identifiers | |
| 206-44-0 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:33083 |
| ChEMBL | ChEMBL355014 |
| ChemSpider | 8800 |
| ECHA InfoCard | 100.005.376 |
| KEGG | C19425 |
| UNII | 360UOL779Z |
| |
| |
| Properties | |
| C16H10 | |
| Molar mass | 202.26 g/mol |
| Appearance | Yellow to green needles |
| Density | 1.252 g/cm3 (0 °C), solid |
| Melting point | 110.8 °C (231.4 °F; 383.9 K) |
| Boiling point | 375 °C (707 °F; 648 K) |
| 265 μg/l (25 °C) | |
| Viscosity | 0.652 cP at 20 °C |
| Structure | |
| Planar | |
| 0.34 D | |
| Hazards | |
| Flash point | 210 °C (410 °F; 483 K) |
| ? °C | |
| Related compounds | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Fluoranthene is a polycyclic aromatic hydrocarbon (PAH). The molecule can be viewed as the fusion of naphthalene and benzene unit connected by a five-membered ring. Although samples are often pale yellow, the compound is colorless. It is soluble in nonpolar organic solvents.[1] It is a member of the class of PAHs known as non-alternant PAHs because it has rings other than those with six carbon atoms. It is a structural isomer of the alternant PAH pyrene. It is not as thermodynamically stable as pyrene. Its name is derived from its fluorescence under UV light. It is used in the synthesis of Florantyrone.
Occurrence
Coal tar
Fluoranthene was originally isolated from coal tar pitch. It is still obtained from the high boiling fraction of coal tar, representing a few percent by weight.[1]
Pollutant
Fluoranthene is found in many combustion products, along with other PAHs. Its presence is an indicator of less efficient or lower-temperature combustion, since non-alternant PAHs are less preferred in formation than alternant PAHs. Fluoranthene is one of the U.S. Environmental Protection Agency's 16 priority pollutant PAHs. Fluoranthene has been classified by the International Agency for Research on Cancer as a group 3 carcinogen, "not classifiable as to its carcinogenicity to humans"[1] , however it was found to possess carcinogenic properties in case of newborn mice according to short-term lung tumor assay (Busby et al., 1984).[2]
Interstellar medium
In February 2014, NASA announced a greatly upgraded database for tracking polycyclic aromatic hydrocarbons (PAHs), including fluoranthene, in the universe. According to scientists, more than 20% of the carbon in the universe may be associated with PAHs, possible starting materials for the formation of life. PAHs seem to have been formed shortly after the Big Bang, are widespread throughout the universe, and are associated with new stars and exoplanets.[3]
References
- 1 2 Karl Griesbaum, Arno Behr, Dieter Biedenkapp, Heinz-Werner Voges, Dorothea Garbe, Christian Paetz, Gerd Collin, Dieter Mayer, Hartmut Höke “Hydrocarbons” in Ullmann's Encyclopedia of Industrial Chemistry 2002 Wiley-VCH, Weinheim. doi:10.1002/14356007.a13_227
- ↑ http://cira.ornl.gov/documents/FLUORANT.pdf
- ↑ Hoover, Rachel (February 21, 2014). "Need to Track Organic Nano-Particles Across the Universe? NASA's Got an App for That". NASA. Retrieved February 22, 2014.
External links
- National Pollutant Inventory - Polycyclic Aromatic Hydrocarbon Fact Sheet
- Chemical Profile for FLUORANTHENE from Scorecard
- FLUORANTHENE Chemical Fact Sheet by Spectrum Laboratories
- TOXICITY SUMMARY FOR FLUORANTHENE August 1993