HLA-A11
| HLA-A11 (MHC Class I, A cell surface antigen) | |||
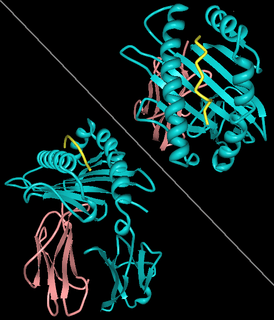 | |||
| Rendering of A11 (A*1101-B2M) in complex with HBV peptide homologue2HN7 HLA-A11 'alpha chain' (Cyan), β2-microglobulin (Rose), HBV peptide (yellow). | |||
| - | |||
| Protein | transmembrane receptor/ligand | ||
| Structure | αβ heterodimer | ||
| Subunits | HLA-A*11--, β2-microglobulin | ||
| Older names | "HL-A11" | ||
| - | |||
| subtype | allele | Available structures | |
| A11E, A11.1 | *1101 | 2hn7, 1x7q, 1qvo, 1q94 | |
| A11K, A11.2 | *1102 | ||
| rare alleles | |||
| A11.3 | *1103 | ||
| A11.4 | *1104 | ||
| Alleles link-out to IMGT/HLA database at EBI | |||
HLA-A11 (A11) is a human leukocyte antigen serotype within HLA-A "A" serotype group. The serotype is determined by the antibody recognition of α11 subset of HLA-A α-chains. For A11, the alpha "A" chain are encoded by the HLA-A*11 allele group and the β-chain are encoded by B2M locus.[1] This group currently is dominated by A*1101. A11 and A*11 are almost synonymous in meaning. A11 is more common in East Asia than elsewhere, it is part of a several long haplotypes that appear to have been frequent in the ancient peoples of Asia.[2]
Serotype
| A*11 | A11 | Sample |
| allele | % | size (N) |
| *1101 | 99 | 2530 |
| *1102 | 76 | 42 |
| *1103 | 83 | 18 |
| *1104 | 60 | 6 |
Serotyping of A11 demonstrates better recognition of the *1101 gene products and poorer recognition of other A*11 gene products. There are ~40 recognized alleles of A*11. There is only one null classified as A11.
In infectious disease
Associations have been observed between A11 and familial otosclerosis,[4][5] pulmonary tuberculosis,[6] leprosy,[7] and cytomegalovirus infection with epilepsy.[8] These and other studies suggest an involvement between A11 and secondary effects of certain herpes virus infections. A11 was also found increase in supraglottic cancer with poor 3 year survival.[9] In osteosarcoma A11 was found elevated.[10]
There is a strong association between anti-depressant induced hepatitis and HLA-A11.[11] In autoimmune hepatitis, A11 has a synergistic effect, acting together with DR4 and DR3 to increase the odds of disease to over 300.[12]
A11 is also part of a haplotype A11-Cw4-B35-DR1-DQ1 that is a second factor in the rapid progression of HIV.[13] The involvement of non-Hodgkin's lymphoma primarily as a result of Epstein-Barr virus reinfection does not appear to be a cause in this acceleration.[14]
Epstein-Barr Virus anomaly
There are at least a couple of forms of lymphoproliferative diseases that appear to arise from unresolved Epstein-Barr virus infection. Examination of the virus itself has led to the discovery of strains that can all but turn off the A11-mediated class I response to the virus in A11 enriched peoples (see tables below). This ability to turn off the immune system and for the virus to remain active is a factor in carcinogenesis. Early studies of A serotypes revealed and association of A11 with Hodgkin's lymphoma and recent studies have shown a complex involvement of Epstein-Barr virus infection as a consequence of low A11 control over infection.[15][16]
Burkitt's lymphoma eventually lead to the discovery of the virus, however this disease is more evident in Africa. An involvement in cytotoxic T-lymphocytes down-regulation in Burkitt's lymphoma was subsequently discovered,[17][18] More recent studies show A11 is down-regulated, and that other genetic defects are a likely cause.[19] The ability to present EB virus antigens revealed a defect in the process after antigen process but before TAP1 involvement.[20] Other studies indicated that peptides bind A11 in delivery to the cell surface for CTL screening, but fall off, and are destroyed intracellularly.[21] However, A3 and A11 can process and load antigens even when proteosome activity is diminished suggesting an alternative mechanism for loading which may benefit in recovery from some disease but impair recovery of others.
It appears that these and other viruses have learned to exploit some defect in the region surrounding A11 that allows the near complete shut-down of gene expression. Oddly, in Africa A11 is at very low frequencies, and homozygotes are rare, suggesting that other genetic susceptibilities may exist that steer the virus toward Burkitt's lymphoma.
Alleles
| Study population |
Freq. (in %)[22] |
|---|---|
| Papua New Guinea Madang | 63.6 |
| PNG West Schrader Ranges | 55.0 |
| Taiwan Hakka | 40.0 |
| PNG Wosera | 38.5 |
| ChinaYunnan Naxi | 38.0 |
| Taiwan Tao | 36.0 |
| China Guangxi Maonan | 35.2 |
| China Guangzhou | 33.8 |
| Taiwan Minnan (1) | 30.9 |
| Thailand | 29.9 |
| China Wuhan | 29.3 |
| Taiwan Pazeh | 28.2 |
| China South Han | 27.7 |
| Thailand Northeast | 27.1 |
| Singapore Chinese | 26.5 |
| Taiwanese of Middle China… | 26.2 |
| PNG New Britain Rabaul | 26.0 |
| Pakistan Brahui | 25.2 |
| Australia Indig. Groote E… | 24.0 |
| India New Delhi | 23.5 |
| USA Asian | 23.0 |
| Pakistan Baloch | 22.2 |
| Taiwan Thao | 21.7 |
| China, Shandong, Linqu Co… | 20.4 |
| Pakistan Sindhi | 19.6 |
| Australia Indig. Cape Yor… | 18.0 |
| PNG Karimui Plateau | 17.9 |
| Singapore Riau Malay | 17.7 |
| Taiwan Siraya | 17.6 |
| China Beijing | 16.4 |
| Pakistan Pathan | 16.3 |
| China Inner Mongolia | 16.2 |
| American Samoa | 16.0 |
| Spain Basque Arratia Vall… | 16.0 |
| China Qinghai Hui | 15.9 |
| Russia Murmansk Saomi | 14.0 |
| Singapore Javanese Indone… | 14.0 |
| New Caledonia | 13.1 |
| Japan Aichi | 12.7 |
| Georgia Svaneti Svans | 12.5 |
| India North Hindus | 12.5 |
| Pakistan Burusho | 12.5 |
| China Yunnan Han (2) | 12.3 |
| India Mumbai Marathas | 12.3 |
| China Harbin N. Korean | 12.1 |
| India Andhra Pradesh Goll… | 11.9 |
| Taiwan Saisiat | 11.8 |
| Oman | 11.4 |
| Israel Arab Druse | 11.0 |
| South Korea (3) | 10.8 |
| Taiwan Tsou | 10.8 |
| Mongolia Khoton Tarialan | 10.7 |
| Sri Lanka Colombo Sinhale… | 10.4 |
| Georgia Tibilisi Kurds | 10.0 |
| Mongolia Khalkha | 10.0 |
| Australia Indig. Kimberly | 9.7 |
| India North Delhi | 9.4 |
| Japan Hyogo | 9.4 |
| Spain North Cabuernigo | 8.9 |
| Saudi Arabia | 8.7 |
| Russia Tuva (2) | 8.7 |
| Spain North Cantabrian | 8.4 |
| Japan Central | 8.2 |
| Romanian | 8.2 |
| Greece North | 8.0 |
| Ireland Northern | 8.0 |
| Taiwan Atayal | 8.0 |
| Jordan Amman | 7.9 |
| Italy North (1) | 7.7 |
| Italy Sardinia (3) | 7.7 |
| Australia Indig. Yuendumu | 7.6 |
| Mongolia Tsaatan | 7.6 |
| USA Caucasians (3) | 7.4 |
| Bulgaria | 7.3 |
| Taiwan Taroko | 7.3 |
| Philippines Ivatan | 7.0 |
| USA Caucasian Bethesda | 7.0 |
| Portugal Centre (2) | 6.8 |
| Australia New South Wales | 6.7 |
| Morocco | 6.7 |
| USA Hawaii Okinawa | 6.7 |
| Wales | 6.6 |
| Serbia | 6.5 |
| Georgia Tibilisi Georgian… | 6.2 |
| Ireland South | 6.2 |
| Algeria(1) | 6.2 |
| Brazil | 6.1 |
| Spain Basque Gipuzkoa Pro… | 6.1 |
| Turkey (2) | 6.1 |
| Russia Northwest | 6.0 |
| Portugal South pop2 | 5.9 |
| England Newcastle | 5.7 |
| Italy North Pavia | 5.6 |
| Israeli Jews | 5.4 |
| Sweden Stockholm | 5.1 |
| Finland | 5.0 |
| Macedonia (4) | 4.9 |
| Italy Bergamo | 4.8 |
| Morocco Berber Nador Meta… | 4.8 |
| Allele frequencies presented, only | |
| Study population |
Freq. (in %)[22] |
|---|---|
| Taiwan Saisiat | 12.7 |
| Taiwan Pazeh | 10.9 |
| Taiwan Ami | 8.7 |
| Taiwan Puyuma | 7.0 |
| Taiwan Siraya | 6.9 |
| China Guangxi Maonan | 6.5 |
| Taiwan Atayal | 5.2 |
| Philippines Ivatan | 5.0 |
| Ch. Guangdong Meizhou Han | 4.6 |
| Taiwan Hakka | 4.5 |
| Hong Kong Chinese | 4.0 |
| Taiwan Minnan (1) | 3.9 |
| Thailand | 3.5 |
| Singapore Chinese | 3.0 |
| Taiwan Tao | 3.0 |
| Taiwan Tsou | 2.9 |
| China Beijing Shijiazhuan… | 2.0 |
| Italy North pop 1 | 1.9 |
| Taiwan Taroko | 1.8 |
| China Inner Mongolia | 1.0 |
| India West Coast Parsis | 1.0 |
| Taiwan Bunun | 1.0 |
| China Qinghai Hui | 0.9 |
| China Yunnan Lisu | 0.6 |
| Japan (3) | 0.2 |
| Allele frequencies presented, only | |
| Study population |
Freq. (in %)[22] |
|---|---|
| China Yunnan Lisu | 5.1 |
| China Yunnan Nu | 3.8 |
| China Beijing Shijiazhuan… | 0.2 |
| Allele frequencies presented, only | |
| Study population |
Freq. (in %)[22] |
|---|---|
| Georgia Svaneti Svans | 1.3 |
| Singapore Riau Malay | 1.2 |
| American Samoa | 1.0 |
| India North Hindus | 1.0 |
| Israel Ashkenazi and Non … | 0.4 |
| China Beijing Shijiazhuan… | 0.2 |
| Allele frequencies presented, only | |
Disease Associations
A*1104 is associated with increased risk for cervical neoplasia resulting from human papillomavirus infection[23]
A11-B Haplotypes
A11-B13
- A11-Cw2-B13 (Li)
- A11-Cw9-B13 (Southern China & SE Asia)
- A11-C10-B13 Buyi
- A11-CBL-B13 Northern China
References
- ↑ Arce-Gomez B, Jones EA, Barnstable CJ, Solomon E, Bodmer WF (February 1978). "The genetic control of HLA-A and B antigens in somatic cell hybrids: requirement for beta2 microglobulin". Tissue Antigens. 11 (2): 96–112. doi:10.1111/j.1399-0039.1978.tb01233.x. PMID 77067.
- ↑ de Campos-Lima PO, Levitsky V, Brooks J, et al. (April 1994). "T cell responses and virus evolution: loss of HLA A11-restricted CTL epitopes in Epstein-Barr virus isolates from highly A11-positive populations by selective mutation of anchor residues". J. Exp. Med. 179 (4): 1297–305. doi:10.1084/jem.179.4.1297. PMC 2191457
 . PMID 7511684.
. PMID 7511684. - ↑ Allele Query Form IMGT/HLA - European Bioinformatics Institute
- ↑ Gregoriadis S, Zervas J, Varletzidis E, Toubis M, Pantazopoulos P, Fessas P (December 1982). "HLA antigens and otosclerosis. A possible new genetic factor". Arch Otolaryngol. 108 (12): 769–71. doi:10.1001/archotol.1982.00790600013004. PMID 6983341.
- ↑ Singhal SK, Mann SB, Datta U, Panda NK, Gupta AK (1999). "Genetic correlation in otosclerosis". Am J Otolaryngol. 20 (2): 102–5. doi:10.1016/S0196-0709(99)90019-4. PMID 10203160.
- ↑ Xu XP, Li SB, Wang CY, Li QH (June 1986). "Study on the association of HLA with pulmonary tuberculosis". Immunol. Invest. 15 (4): 327–32. doi:10.3109/08820138609052951. PMID 3759149.
- ↑ Kim SJ, Choi IH, Dahlberg S, Nisperos B, Kim JD, Hansen JA (March 1987). "HLA and leprosy in Koreans". Tissue Antigens. 29 (3): 146–53. doi:10.1111/j.1399-0039.1987.tb01567.x. PMID 3603547.
- ↑ Iannetti P, Morellini M, Raucci U, Cappellacci S (1988). "HLA antigens, epilepsy and cytomegalovirus infection". Brain Dev. 10 (4): 256–8. doi:10.1016/s0387-7604(88)80008-1. PMID 2851270.
- ↑ Konieczna A, Turowski G (1993). "HLA-ABC antigens in supraglottic cancer patients and their relationship with incidence and survival". Mater Med Pol. 25 (2): 73–9. PMID 8072312.
- ↑ Barona P, Sierrasesúmaga L, Antillón F, Villa-Elízaga I (1993). "Study of HLA antigens in patients with osteosarcoma". Hum. Hered. 43 (5): 311–4. doi:10.1159/000154149. PMID 8406520.
- ↑ Berson A, Fréneaux E, Larrey D, et al. (March 1994). "Possible role of HLA in hepatotoxicity. An exploratory study in 71 patients with drug-induced idiosyncratic hepatitis". J. Hepatol. 20 (3): 336–42. doi:10.1016/S0168-8278(94)80004-9. PMID 8014443.
- ↑ Marcos Y, Fainboim HA, Capucchio M, et al. (June 1994). "Two-locus involvement in the association of human leukocyte antigen with the extrahepatic manifestations of autoimmune chronic active hepatitis". Hepatology. 19 (6): 1371–4. doi:10.1016/0270-9139(94)90230-5. PMID 8188167.
- ↑ Roger M (June 1998). "Influence of host genes on HIV-1 disease progression". FASEB J. 12 (9): 625–32. PMID 9619442.
- ↑ Chu PG, Chang KL, Chen WG, et al. (September 1999). "Epstein-Barr virus (EBV) nuclear antigen (EBNA)-4 mutation in EBV-associated malignancies in three different populations". Am. J. Pathol. 155 (3): 941–7. doi:10.1016/S0002-9440(10)65193-0. PMC 1866909
 . PMID 10487851.
. PMID 10487851. - ↑ Forbes JF, Morris PJ (May 1972). "Analysis of HL-A antigens in patients with Hodgkin's disease and their families". J. Clin. Invest. 51 (5): 1156–63. doi:10.1172/JCI106908. PMC 292245
 . PMID 5020429.
. PMID 5020429. - ↑ Dolcetti R, Frisan T, Sjöberg J, et al. (August 1995). "Identification and characterization of an Epstein-Barr virus-specific T-cell response in the pathologic tissue of a patient with Hodgkin's disease". Cancer Res. 55 (16): 3675–81. PMID 7627978.
- ↑ Masucci MG, Torsteindottir S, Colombani J, Brautbar C, Klein E, Klein G (July 1987). "Down-regulation of class I HLA antigens and of the Epstein-Barr virus-encoded latent membrane protein in Burkitt lymphoma lines". Proc. Natl. Acad. Sci. U.S.A. 84 (13): 4567–71. doi:10.1073/pnas.84.13.4567. PMC 305131
 . PMID 3037521.
. PMID 3037521. - ↑ Gavioli R, De Campos-Lima PO, Kurilla MG, Kieff E, Klein G, Masucci MG (July 1992). "Recognition of the Epstein-Barr virus-encoded nuclear antigens EBNA-4 and EBNA-6 by HLA-A11-restricted cytotoxic T lymphocytes: implications for down-regulation of HLA-A11 in Burkitt lymphoma". Proc. Natl. Acad. Sci. U.S.A. 89 (13): 5862–6. doi:10.1073/pnas.89.13.5862. PMC 49397
 . PMID 1321426.
. PMID 1321426. - ↑ Imreh MP, Zhang QJ, de Campos-Lima PO, et al. (July 1995). "Mechanisms of allele-selective down-regulation of HLA class I in Burkitt's lymphoma". Int. J. Cancer. 62 (1): 90–6. doi:10.1002/ijc.2910620117. PMID 7601573.
- ↑ Frisan T, Zhang QJ, Levitskaya J, Coram M, Kurilla MG, Masucci MG (October 1996). "Defective presentation of MHC class I-restricted cytotoxic T-cell epitopes in Burkitt's lymphoma cells". Int. J. Cancer. 68 (2): 251–8. doi:10.1002/(SICI)1097-0215(19961009)68:2<251::AID-IJC19>3.0.CO;2-D. PMID 8900437.
- ↑ Levitsky V, Zhang QJ, Levitskaya J, Kurilla MG, Masucci MG (December 1997). "Natural variants of the immunodominant HLA A11-restricted CTL epitope of the EBV nuclear antigen-4 are nonimmunogenic due to intracellular dissociation from MHC class I:peptide complexes". J. Immunol. 159 (11): 5383–90. PMID 9548478.
- 1 2 3 4 Middleton D, Menchaca L, Rood H, Komerofsky R (2003). "New allele frequency database: http://www.allelefrequencies.net". Tissue Antigens. 61 (5): 403–7. doi:10.1034/j.1399-0039.2003.00062.x. PMID 12753660.
- ↑ Chan D, Cheung T, Tam A, Cheung J, Yim S, Lo K, Siu N, Zhou D, Chan P (2005). "Risk association between human leukocyte antigen-A allele and high-risk human papillomavirus infection for cervical neoplasia in Chinese women.". J Infect Dis. 192 (10): 1749–56. doi:10.1086/497342. PMID 16235173.