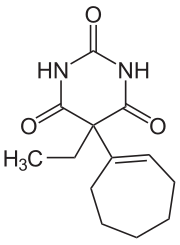
Heptabarb
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral[1] |
| ATC code | N05CA11 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 83%[1] |
| Metabolism | Hepatic |
| Biological half-life | 6.1-11.2 hours[1] |
| Excretion | Renal[1] |
| Identifiers | |
| |
| Synonyms | G-475 |
| CAS Number | 509-86-4 |
| PubChem (CID) | 10518 |
| DrugBank |
DB01354 |
| ChemSpider |
10081 |
| UNII |
V10R70ML23 |
| KEGG |
C17725 |
| ChEMBL |
CHEMBL468837 |
| ECHA InfoCard | 100.007.371 |
| Chemical and physical data | |
| Formula | C13H18N2O3 |
| Molar mass | 250.294 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Heptabarb (INN; Eudan, Medapan, Medomin, Noctyn), also known as heptabarbitone (BAN) or heptabarbital, is a sedative and hypnotic drug of the barbiturate family.[2][3] It was used in Europe for the treatment of insomnia from the 1950s onwards, but has since been discontinued.[2][3]
See also
References
- 1 2 3 4 Breimer DD, de Boer AG (December 1975). "Pharmacokinetics and relative bioavailability of heptabarbital and heptabarbital sodium after oral administration to man". European Journal of Clinical Pharmacology. 9 (2-3): 169–78. doi:10.1007/bf00614014. PMID 9299.
- 1 2 C. R Ganellin; D. J Triggle; F.. Macdonald (1997). Dictionary of pharmacological agents. CRC Press. p. 1003. ISBN 978-0-412-46630-4. Retrieved 26 November 2011.
- 1 2 Index nominum 2000: international drug directory. Taylor & Francis US. 2000. p. 513. ISBN 978-3-88763-075-1. Retrieved 26 November 2011.
This article is issued from Wikipedia - version of the 10/11/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.