Infectious bursal disease
| Infectious bursal disease | |
|---|---|
| Virus classification | |
| Group: | Group III (dsRNA) |
| Family: | Birnaviridae |
| Genus: | Avibirnavirus |
| Species | |
|
Infectious bursal disease | |
Infectious bursal disease (also known as IBD, Gumboro Disease, Infectious Bursitis and Infectious Avian Nephrosis) is a highly contagious disease of young chickens caused by infectious bursal disease virus (IBDV),[1] characterized by immunosuppression and mortality generally at 3 to 6 weeks of age. The disease was first discovered in Gumboro, Delaware in 1962. It is economically important to the poultry industry worldwide due to increased susceptibility to other diseases and negative interference with effective vaccination. In recent years, very virulent strains of IBDV (vvIBDV), causing severe mortality in chicken, have emerged in Europe, Latin America, South-East Asia, Africa and the Middle East. Infection is via the oro-fecal route, with affected bird excreting high levels of the virus for approximately 2 weeks after infection.
Virology
IBDV is a double stranded RNA virus that has a bi-segmented genome and belongs to the genus Avibirnavirus of family Birnaviridae. There are two distinct serotypes of the virus, but only serotype 1 viruses cause disease in poultry.[2] At least six antigenic subtypes of IBDV serotype 1 have been identified by in vitro cross-neutralization assay. Viruses belonging to one of these antigenic subtypes are commonly known as variants, which were reported to break through high levels of maternal antibodies in commercial flocks, causing up to 60 to 100 percent mortality rates in chickens. With the advent of highly sensitive molecular techniques, such as reverse transcription polymerase chain reaction (RT-PCR) and restriction fragment length polymorphism (RFLP), it became possible to detect the vvIBDV, to differentiate IBDV strains, and to use such information in studying the molecular epidemiology of the virus.
IBDV genome consists of two segments, A and B, which are enclosed within a nonenveloped icosahedral capsid.[3] The genome segment B (2.9 kb) encodes VP1, the putative viral RNA polymerase. The larger segment A (3.2 kb) encodes viral proteins VP2, VP3, VP4, and VP5. Among them, VP2 protein contains important neutralizing antigenic sites and elicits protective immune response and most of the amino acid (AA) changes between antigenically different IBDVs are clustered in the hypervariable region of VP2. Thus, this hypervariable region of VP2 is the obvious target for the molecular techniques applied for IBDV detection and strain variation studies.
Viral structure
The IBDV capsid protein exhibits structural domains that show homology to those of the capsid proteins of some positive-sense single-stranded RNA viruses, such as the nodaviruses and tetraviruses, as well as the T=13 capsid shell protein of the Reoviridae. The T=13 shell of the IBDV capsid is formed by trimers of VP2, a protein generated by removal of the C-terminal domain from its precursor, pVP2. The trimming of pVP2 is performed on immature particles as part of the maturation process. The other major structural protein, VP3, is a multifunctional component lying under the T=13 shell that influences the inherent structural polymorphism of pVP2. The virus-encoded RNA-dependent RNA polymerase, VP1, is incorporated into the capsid through its association with VP3. VP3 also interacts extensively with the viral dsRNA genome.[1]
Pathogenesis
Clinical disease is associated to bird age with the greatest bursal mass, which occurs between 3 and 6 weeks of age. The greatest bursal mass is mostly a result of a large population of maturing IgM-bearing B-lymphocytes (lymphoblasts), the main target of infection. Young birds at around two to eight weeks of age that have highly active bursa of Fabricius are more susceptible to disease. Birds over eight weeks are resistant to challenge and will not show clinical signs unless infected by highly virulent strains.
Subclinical disease occurs in chickens infected before three weeks of age. At this age the B-lymphoblast population is smaller and the systemic effects are insufficient for generating clinical signs. However, the B-cell destruction is usually most severe in subclinically infected young, as virus will destroy a smaller population and most cells in one place (the bursa).
After ingestion, the virus destroys the lymphoid follicles in the bursa of Fabricius as well as the circulating B-cells in the secondary lymphoid tissues such as GALT (gut-associated lymphoid tissue), CALT (conjunctiva), BALT (Bronchial) caecal tonsils, Harderian gland, etc. Acute disease and death is due to the necrotizing effect of these viruses on the host tissues. Kidney failure is a common cause of mortality. If the bird survives and recovers from this phase of the disease, it remains immunocompromised which means it is more susceptible to other diseases.
Clinical signs
Disease may appear suddenly and morbidity typically reaches 100%. In the acute form birds are prostrated, debilitated and dehydrated. They produce a watery diarrhea and may have swollen feces-stained vent. Most of the flock is recumbent and have ruffled feathers. Mortality rates vary with virulence of the strain involved, the challenge dose, previous immunity, presence of concurrent disease, as well as the flock's ability to mount an effective immune response. Immunosuppression of very young chickens, less than three weeks of age, is possibly the most important outcome and may not be clinically detectable (subclinical). In addition, infection with less virulent strains may not show overt clinical signs, but birds that have bursal atrophy with fibrotic or cystic follicles and lymphocytopenia before six weeks of age, may be susceptible to opportunistic infection and may die of infection by agents that would not usually cause disease in immunocompetent birds.
Diagnosis
A preliminary diagnosis can usually be made based on flock history, clinical signs and post-mortem (necropsy) examinations. However, definitive diagnosis can only be achieved by the specific detection and/or isolation and characterization of IBDV. Immunofluorescence or immunohistochemistry tests, based on anti-IBDV labelled antibodies, or in-situ hybridization, based on labelled complementary cDNA sequence probe, are useful for the specific detection of IBDV in infected tissues. RT-PCR (as mentioned above) was designed for the detection of IBDV genome, such as VP1 coding gene, with the possibility of PCR product sequences be determined for genetically comparing isolates and producing phylogenetic trees. Serological tests such as agar gel precipitation and ELISA, for detecting antibodies, are used for monitoring vaccine responses and might be additional information for diagnosis of infection of unvaccinated flocks.
Necropsy examination will usually show changes in the bursa of Fabricius such as swelling, oedema, haemorrhage, the presence of a jelly serosa transudate and eventually, bursal atrophy. Pathological changes, especially haemorrhages, may also be seen in the skeletal muscle, intestines, kidney and spleen.
Treatment & Control
Peri-focal vaccination may not be effective for the combat of an outbreak, due to the rapidity of wild-IBDV spreading.
Passive immunity may protect against challenge with homologous IBDV, as does previous infection with homologous avirulent strains. Breeder flocks may be immunised against IBD so that they would transfer protective antibodies to their progenies, such as broiler and pullet chicks. Low-attenuated vaccine strains may cause damage to the bursa of Fabricius and immunosuppression in susceptible chicks. Biosecurity with adequate restriction to farm visitation and distancing from other flocks. Post outbreak hygiene measures may not be effective as the virus can survive for long periods in both housing and water.
-
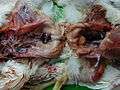
Two enlarged bursae: yellowish grey (right) and haemorrhagic (left)
-
Caseous exudate in bursa of Fabricius
-
Lesions of kidneys
-
Haemorrhages in proventriculus and gizzard
See also
| Wikimedia Commons has media related to Infectious bursal disease. |
References
- 1 2 Caston; et al. (2008). "Infectious Bursal Disease Virus (IBDV)". Segmented Double-stranded RNA Viruses: Structure and Molecular Biology. Caister Academic Press. ISBN 978-1-904455-21-9.
- ↑ "Infectious Bursal Disease: Introduction". The Merck Veterinary Manual. 2006. Retrieved 2007-06-26.
- ↑ ICTVdB Management (2006). 00.009.0.02.001. Infectious bursal disease virus. In: ICTVdB - The Universal Virus Database, version 4. Büchen-Osmond, C. (Ed), Columbia University, New York, USA
External links
- Description, with photo of diseased organ
- IBD in Commercial Broilers
- Infectious Bursal Disease
- Infectious Bursal Disease, expert reviewed and published by WikiVet at http://en.wikivet.net/Infectious_Bursal_Disease, accessed 13/09/2011