Lyman continuum photons
Lyman continuum photons (abbrev. LyC), shortened to Ly continuum photons or Lyc photon, are the photons emitted from stars at energies above the Lyman limit. Hydrogen is ionized by absorbing LyC. Working off of Victor Schumann's discovery of ultraviolet light, from 1906 to 1914, Theodore Lyman observed that atomic hydrogen absorbs light only at specific frequencies (or wavelengths) and the Lyman series is thus named after him.[1][2] All the wavelengths in the Lyman series are in the ultraviolet band. This quantized absorption behavior occurs only up to an energy limit, known as the ionization energy. In the case of neutral atomic hydrogen, the minimum ionization energy is equal to the Lyman limit, where the photon has enough energy to completely ionize the atom, resulting in a free proton and a free electron. Above this energy (below this wavelength), all wavelengths of light may be absorbed. This forms a continuum in the energy spectrum; the spectrum is continuous rather than composed of many discrete lines, which are seen at lower energies.[3][4]
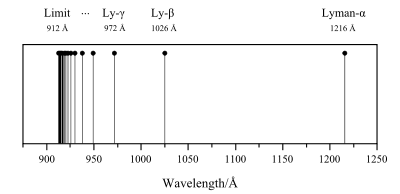
The Lyman limit is at the wavelength of 91.2 nm (912 Å), corresponding to a frequency of 3.29 million GHz and an energy of 13.6 eV.[3] LyC energies are mostly in the ultraviolet C portion of the electromagnetic spectrum (see Lyman series). Although X-rays and gamma-rays will also ionize a hydrogen atom, there are far fewer of them emitted from a star's photosphere—LyC are predominantly UV-C. The photon absorption process leading to the ionization of atomic hydrogen can occur in reverse: an electron and a proton can collide and form atomic hydrogen. If the two particles were traveling slowly (so that kinetic energy can be ignored), then the photon the atom emits upon its creation will theoretically be 13.6 eV (in reality, the energy will be less if the atom is formed in an excited state). At faster speeds, the excess (kinetic) energy is radiated (but momentum must be conserved) as photons of lower wavelength (higher energy). Therefore, photons with energies above 13.6 eV are emitted by the combination of energetic protons and electrons forming atomic hydrogen, and emission from photoionized hydrogen.
See also
- Balmer limit
- Lyman-alpha blob
- Lyman-alpha forest
- Lyman-break galaxy
- Lyman series
- Haro 11 - One of the two galaxies in the local universe that 'leaks' Lyman continuum photons.
- Tololo-1247-232 - The second galaxy in the local universe that 'leaks' Lyman continuum photons.
- Pea galaxy - At least two Green Peas are suspected of being LyC 'leakers'.
References
- ↑ Lyman, Theodore (1906), "The Spectrum of Hydrogen in the Region of Extremely Short Wave-Length", Memoirs of the American Academy of Arts and Sciences, New Series, 13 (3): 125–146, ISSN 0096-6134, JSTOR 25058084,
In a preliminary paper1 the author has given the wave-length of more than one hundred and thirty lines in the region of the spectrum lying between the values 1850 and 1030 tenth metres. It is the object of the present paper to compare the results obtained by the author with those given by Schumann; to describe the apparatus used in this research and to call attention to some new facts which have come to light since the publication of the first notice.
- ↑ Lyman, Theodore (1914), "An Extension of the Spectrum in the Extreme Ultra-Violet", Nature, 93: 241, Bibcode:1914Natur..93..241L, doi:10.1038/093241a0
- 1 2 Dipankar Bhattacharya (August–December 2003). "Matter and Radiation" (PDF). Fundamentals of Astrophysics. India: Inter-University Centre for Astronomy and Astrophysics.
In most situations, the total emission by the free-free process far exceeds the recombination radiation, but the recombination radiation can introduce characteristic spectral features at ionisation thresholds in the continuum radiation. For Hydrogen, the highest ionisation threshold, called the Lyman Limit, corresponds to an energy of 13.6 eV or an wavelength of 912Å. Recombination radiation from Hydrogen at wavelengths shorter than this is called the “Lyman continuum”.
- ↑ "Lyman limit". A Dictionary of Astronomy. 1997. Archived from the original on May 23, 2011.
Lyman limit The short-wavelength end of the hydrogen Lyman series, at 91.2 nm. It corresponds to the energy required for an electron in the hydrogen ground state to jump completely out of the atom, leaving the atom ionized.