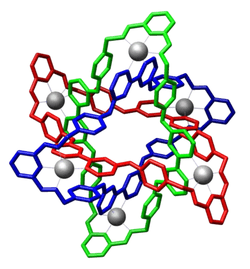
Molecular Borromean rings
Molecular Borromean rings are an example of a mechanically-interlocked molecular architecture in which three macrocycles are interlocked in such a way that breaking any macrocycle allows the others to disassociate. They are the smallest examples of Borromean rings. The synthesis of molecular Borromean rings was reported in 2004 by the group of J. Fraser Stoddart. The so-called Borromeate is made up of three interpenetrated macrocycles formed from the reaction between 2,6-diformylpyridine and diamine compounds, complexed with zinc.[1]
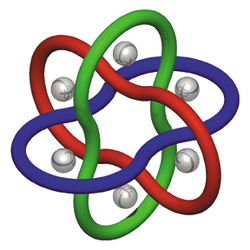 Schematic of a molecular Borromean ring. |
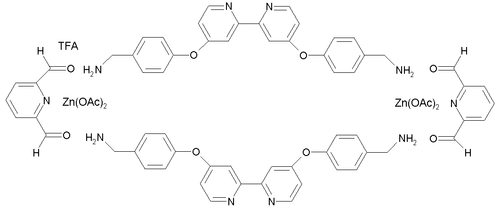
This compound was synthesized from two building blocks: 2,6-diformylpyridine (a pyridine with two aldehyde groups) and a diamine containing a 2,2'-bipyridine group. Zinc acetate is added as the template for the reaction, resulting in one zinc atom in each of a total of 6 pentacoordinate complexation sites. Trifluoroacetic acid (TFA) is added to catalyse the imine bond-forming reactions. The preparation of the tri-ring Borromeate involves a total of 18 precursor molecules and is only possible because the building blocks self-assemble through 12 aromatic pi-pi interactions and 30 zinc to nitrogen dative bonds. Because of these interactions, the Borromeate is thermodynamically the most stable reaction product out of potentially many others. As a consequence of all the reactions taking place being equilibria, the Borromeate is the predominant reaction product.
Reduction with sodium borohydride in ethanol affords the neutral Borromeand.[2] True to a Borromean system, cleavage of just one imine bond (to an amine and an acetal) in this structure breaks the mechanical bond between the three constituent macrocycles, releasing the other two individual rings.
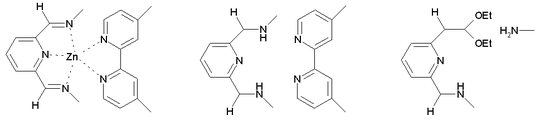
Organic synthesis of this seemingly complex compound is in reality fairly simple; the reason why the Stoddart group has suggested it for a classroom experiment on a gram scale.[3]
See also
References
- 1 2 Molecular Borromean Rings Kelly S. Chichak, Stuart J. Cantrill, Anthony R. Pease, Sheng-Hsien Chiu, Gareth W. V. Cave, Jerry L. Atwood, J. Fraser Stoddart Science, Vol 304, Issue 5675, 1308-1312 , 28 May 2004 Abstract
- ↑ Nanoscale Borromean links for real Andrea J. Peters, Kelly S. Chichak, Stuart J. Cantrill and J. Fraser Stoddart Chemical Communications, 2005, (27), 3394 - 3396 Abstract
- ↑ Making Molecular Borromean Rings. A Gram-Scale Synthetic Procedure for the Undergraduate Organic Lab Cari D. Pentecost, Nicholas Tangchaivang, Stuart J. Cantrill, Kelly S. Chichak, Andrea J. Peters, and J. Fraser Stoddart Vol. 84 No. 5 May 2007 Journal of Chemical Education 855 Abstract
External links
- Three rings in an inseparable union Michel Freemantle Chemical & Engineering News May 31, 2004 Volume 82, Number 22 p. 5 Article
- Borromean chemistry overview website