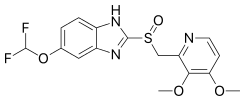
Pantoprazole
 | |
| Clinical data | |
|---|---|
| Trade names | Protonix |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601246 |
| License data |
|
| Pregnancy category | |
| Routes of administration | By mouth and intravenous |
| ATC code | A02BC02 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 77% |
| Metabolism | Liver (CYP2C19) |
| Biological half-life | 1-2 hours |
| Excretion | Urine, Feces |
| Identifiers | |
| |
| CAS Number |
102625-70-7 |
| PubChem (CID) | 4679 |
| IUPHAR/BPS | 7260 |
| DrugBank |
DB00213 |
| ChemSpider |
4517 |
| UNII |
D8TST4O562 |
| KEGG |
D05353 |
| ChEBI |
CHEBI:7915 |
| ChEMBL |
CHEMBL1502 |
| ECHA InfoCard | 100.111.005 |
| Chemical and physical data | |
| Formula | C16H15F2N3O4S |
| Molar mass | 383.371 g/mol |
| 3D model (Jmol) | Interactive image |
| Chirality | Racemic |
| |
| |
| (verify) | |
Pantoprazole, sold under the brand name Protonix among others, is used for short-term treatment of erosive esophagitis associated with gastroesophageal reflux disease (GERD), maintenance of healing of erosive esophagitis, and pathological hypersecretory conditions including Zollinger–Ellison syndrome.[1]
Some common side effects of pantoprazole use in adults include: headache, diarrhea, nausea, abdominal pain, vomiting, flatulence, dizziness, and joint pain (>2%).[1] Use of pantoprazole for a long period of time may lead to chronic inflammation of stomach lining or atrophic gastritis, vitamin B-12 deficiency, and low magnesium.[1][2]
Pantoprazole is a proton pump inhibitor drug that inhibits gastric acid secretion. It works on gastric parietal cells to irreversibly inhibit (H+/K+)-ATPase function and suppress the production of gastric acid.[1][3]
Medical uses
Pantoprazole is used for short-term treatment of erosion and ulceration of the esophagus for adults and pediatric patients 5 years of age and older caused by gastroesophageal reflux disease.[1] It can be used as a maintenance therapy for long-term use after initial response is obtained, but there have not been any controlled studies about the use of pantoprazole past a duration of 12 months.[1] Pantoprazole may also be used in combination with antibiotics to treat ulcers caused by Helicobacter pylori.[4] It can also be used for long-term treatment of Zollinger-Ellison syndrome.[1]
Pregnancy
U.S.A Pregnancy Category B: In reproductive studies using doses largely greater than the recommended doses performed on rats and rabbits, there was no evident harm on the development of the baby.[1]
Breast feeding
Pantoprazole has been found to pass through the breast milk. However, in rodent cancer studies, pantoprazole has been shown to potentially cause tumor growth. The clinical relevance of the finding is unknown, but risks and benefits are recommended for consideration in determining the use of therapy for the mother and child.[1]
Children
Pantoprazole is only indicated for the short-term treatment of erosive esophagitis in children ages 5 and older; and the safety and effectiveness of pantoprazole have only been established in the treatment of erosive esophagitis in children.[1]
Elderly
The incidence of adverse effects occurring in patients aged 65 years and older were similar to those in patients aged 65 years and less.[1]
Adverse effects
- Infection: Stomach acid plays a role in killing ingested bacteria. Use of pantoprazole may increase the chance of developing infections such as pneumonia, particularly in hospitalized patients.[5]
Common
- Gastrointestinal: abdominal pain (6%), diarrhea (9%), flatulence (4%), nausea (7%), vomiting (4%)[1]
- Neurologic: headache (12%), dizziness (3%)[1]
- Neuromuscular and skeletal: arthralgia (3%)[1]
Rare
- Gastrointestinal: constipation, dry mouth, hepatitis[1]
- Blood problems: low white blood cell count, thrombocytopenia[1]
- Immunologic: Stevens-Johnson syndrome, toxic epidermal necrolysis[1]
- Metabolic: elevated creatine kinase, elevated cholesterol levels, elevated liver enzymes (AST/ALT), swelling[1]
- Musculoskeletal: Muscle disorders, bone fracture and infection, Clostridium difficile infection, osteoporosis-related hip fracture, rhabdomyolysis[1]
- Kidneys: interstitial nephritis[6]
- Nutrition: may reduce the absorption of important nutrients, vitamins, and minerals, including certain medications, leaving users at increased risk for pneumonia.[7]
Long-term Use
- Osteoporosis and bone fracture have been observed in patients on high-dose and/or long term (over 1 year) prescription proton pump inhibitors.[8]
- Hypomagnesia has been observed in patients on medications like pantoprazole when taken for longer periods of time (generally 1 year or more, although cases have been reported with regimens as short as 3 months).[2]
Interactions
- Acidity: Due to its effect of reducing stomach acidity, use of pantoprazole can affect absorption of drugs that are pH-sensitive such as ampicillin esters, ketoconazole, atazanavir, iron salts, and mycophenolate moftetil.[1]
Pharmacology

The mechanism of action of pantoprazole is to inhibit the final step in gastric acid production.[1] In the gastric parietal cell of the stomach, pantoprazole covalently binds to the H+/K+ ATP pump to inhibit gastric acid and basal acid secretion.[1] The covalent binding prevents acid secretion of up to 24 hours and longer.[1]
Pantoprazole is metabolized in the liver by the cytochrome P450 system.[9] Metabolism mainly consists of demethylation by CYP2C19 followed by sulfation. Another metabolic pathway is oxidation by CYP3A4. Pantoprazole metabolites are not thought to have any pharmacological significance. It is usually given with a prokinetic drug because of inactivity in the acidic environment of the stomach. Pantoprazole binds irreversibly to H+K+ATPase (proton pumps) to suppress the secretion of acid. Due to irreversible binding of the pumps, new pumps have to be made before acid production can be resumed.[3] The drug's plasma half-life is about 2 hours.[10]
See also
References
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 "Prescribing Info". Protonix package insert. Retrieved November 3, 2015.
- 1 2 Research, Center for Drug Evaluation and. "Drug Safety and Availability - FDA Drug Safety Communication: Low magnesium levels can be associated with long-term use of Proton Pump Inhibitor drugs (PPIs)". www.fda.gov. Retrieved 2015-11-03.
- 1 2 Richardson, Paul; Hawkey, Christopher J.; Stack, Dr William A. (2012-11-29). "Proton Pump Inhibitors". Drugs. 56 (3): 307–335. doi:10.2165/00003495-199856030-00002. ISSN 0012-6667. PMID 9777309.
- ↑ Dammann, Hans-Gerd; Fölsch, Ulrich R.; Hahn, Eckhart G.; Von Kleist, Detlef-Hasso; Klör, Hans-Ulrich; Kirchner, Thomas; Strobel, Sonja; Kist, Manfred (2000-03-01). "Eradication of H. pylori with Pantoprazole, Clarithromycin, and Metronidazole in Duodenal Ulcer Patients: A Head-to-Head Comparison Between Two Regimens of Different Duration". Helicobacter. 5 (1): 41–51. doi:10.1046/j.1523-5378.2000.00006.x. ISSN 1523-5378. PMID 10672051.
- ↑ Herzig, SJ; Doughty, C; Lahoti, S; Marchina, S; Sanan, N; Feng, W; Kumar, S (November 2014). "Acid-suppressive medication use in acute stroke and hospital-acquired pneumonia.". Annals of Neurology. 76 (5): 712–8. doi:10.1002/ana.24262. PMID 25164323.
- ↑ Ricketson, Jeffrey; Kimel, Gil; Spence, James; Weir, Rene (2009-03-03). "Acute allergic interstitial nephritis after use of pantoprazole". Canadian Medical Association Journal. 180 (5): 535–538. doi:10.1503/cmaj.080456. ISSN 0820-3946. PMC 2645468
 . PMID 19255077.
. PMID 19255077. - ↑ [Dr. John Cooke, chair of Methodist Hospital's cardiovascular services] [Houston Chronicle Health Zone dated Thursday, July 11, 2013 chron.com/refluxmeds] (Journal: Circulation)
- ↑ Research, Center for Drug Evaluation and. "Postmarket Drug Safety Information for Patients and Providers - FDA Drug Safety Communication: Possible increased risk of fractures of the hip, wrist, and spine with the use of proton pump inhibitors". www.fda.gov. Retrieved 2015-11-03.
- ↑ Meyer, U A (1996). "Metabolic interactions of the proton-pump inhibitors lansoprazole, omeprazole and pantoprazole with other drugs". European journal of gastroenterology & hepatology. 8 (Suppl 1): S21–25. doi:10.1097/00042737-199610001-00005.
- ↑ Sachs, George; Shin, Jai Moo; Hunt, Richard (2010-10-06). "Novel Approaches to Inhibition of Gastric Acid Secretion". Current Gastroenterology Reports. 12 (6): 437–447. doi:10.1007/s11894-010-0149-5. ISSN 1522-8037. PMC 2974194
 . PMID 20924727.
. PMID 20924727.