Pyrroline
Pyrrolines, also known under the name dihydropyrroles, are three different heterocyclic organic chemical compounds that differ in the position of the double bond. Pyrrolines are formally derived from the aromate pyrrole by hydrogenation. 1-Pyrroline is a cyclic imine, whereas 2-pyrroline and 3-pyrroline are cyclic amines.
 |
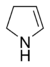 |
 |
| 1-Pyrroline | 2-Pyrroline | 3-Pyrroline |
Substituted pyrrolines
Notable examples of pyrrolines containing various substituents include:
- 2-Acetyl-1-pyrroline, an aroma compound with a white bread-like smell
- Thienamycin, a beta-lactam antibiotic
- MTSL, a chemical used for certain NMR experiments
- Pyrrolysine, an unusual proteinogenic amino acid
- 1-Pyrroline-5-carboxylic acid, a biosynthetic metabolite
- Porphyrin, consisting of two alternating pairs of pyrrol and pyrroline connected via methine (=CH-) bridges
N-substituted pyrrolines can be generated by ring-closing metathesis.[1]
See also
- Pyrrole, the aromatic analog with two double bonds
- Pyrrolidine, the fully saturated analog without double bonds
References
- ↑ Marcelle L. Ferguson, Daniel J. O'leary, And Robert H. Grubbs "Ring-closing Metathesis Synthesis Of N-boc-3-pyrroline" Org. Synth. 2003, volume 80, p. 85. doi:10.15227/orgsyn.080.0085
External links
- Pyrroline, 1-pyrroline, 2-pyrroline, and 3-pyrroline at EMBL-EBI
This article is issued from Wikipedia - version of the 1/31/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.