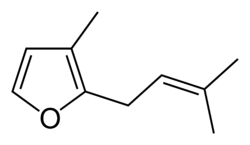
Rosefuran
 | |
| Names | |
|---|---|
| IUPAC name
3-Methyl-2-(3-methyl-2-buten-1-yl)furan | |
| Other names
3-methyl-2-prenylfuran; 2-(3-Methyl-2-butenyl)-3-methylfuran | |
| Identifiers | |
| 15186-51-3 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 76521 |
| PubChem | 84825 |
| |
| |
| Properties | |
| C10H14O | |
| Molar mass | 150.22 g·mol−1 |
| Appearance | colorless liquid |
| Density | 0.9089 @30 °C |
| Boiling point | 103 to 104 °C (217 to 219 °F; 376 to 377 K) at 50 mmHg |
| Hazards | |
| Flash point | 62.22 °C; 144.00 °F; 335.37 K |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Rosefuran (3-methyl-2-prenylfuran) is a liquid boiling at 103-104 °C (at 50 mm Hg), with a density of 0.9089 g/cm3 (at 30 °C), less than that of water.[1] It is an aroma chemical which is a minor constituent of the aroma of the rose (Rosa damascene). Rosefuran is a 2,3-disubstituted furan (3-methyl-2-(3-methyl-2-buten-1-yl)furan). It has an odor threshold of 200 ppb and constitutes 0.16% of Bulgarian rose oil.[2] Rosefuran has been established as a female sex pheromone of an acarid mite, Caloglyphus sp. Concentrations of less than 100 ng of synthetic rosefuran caused sexual excitation in males of the species.[3]
Synthesis
Rosefuran was synthesized in 1968 by George Büchi via the organomercury compound bis(3-methyl-2-furyl)mercury.[4] Rosefuran has been synthesized in many steps from common starting materials starting with prenyl chloride (3-methyl-but-2-en-1-yl chloride).[5] Rosefuran has also been synthesized via cyclization of suitable acyclic precursors, such as, 5-oxogeraniol and 5-hydroxycitral (4-hydoxy-3,7-dimethyl-(E)-2,6-octadienol).[6] Rosefuran has also been prepared via palladium catalyzed cross coupling of the methyl ester of 4-bromo-5-methyl-2-furancarboxylic acid followed by decarboxylation.[7]
Alternatively, rosefuran has been prepared from other furan derivatives. Thus, Wittig reaction of 3-methyl-2-furanacetaldehyde with triphenylphosphonium isopropylide gave rosefuran in 67% yield. The aldehyde was obtained via Claisen rearrangement of 3-(vinyloxymethyl)furan.[8] Similarly, the readily available 3-bromofuran was reacted with 3,3-dimethylallyl bromide and lithium diisopropylamide in THF solution, followed by reaction at with methyl iodide and N-butyllithium in THF, yielding rosefuran.[9]
A patented process for the manufacture of rosefuran via condensation of 3-formylpropionic acid methyl ester with crotonaldehyde has been described. The intermediate ketoaldehyde is cyclized to a furan derivative which is converted to rosefuran in 33% overall yield via Grignard reaction to give 2-methyl-4-(3-methyl-2-furanyl)-2-butanol followed by dehydration.[10]
Applications
Rosefuran is a desirable component of rose oil, a natural fragrance material.[11] Rosefuran is the major constituent (58% by weight) of the essential oil of Perilla ocimoides, a herbaceous, annual plant cultivated in Bangladesh and used as a component of a common spice.[12] Additionally, synthetic rosefuran is recommended (average recommended level in parentheses) for use as a flavor, fragrance chemical for a wide variety of applications including baked goods (5 ppm), breakfast cereal (2 ppm), cheese (3 ppm), condiments (2 ppm), frozen dairy (3 ppm), soups (2 ppm) and snack foods (5 ppm).[13]
References
- ↑ B. Sanjiva Rao; Subramaniam, K. S. (1936). "The occurrence of furan derivatives in volatile oils. III. β-Clausenan and γ-clausenan". Proceedings - Indian Academy of Sciences, Section A. V3A (4): 31–37.
- ↑ Gunther Ohloff (1978). "Importance of minor components in flavors and fragrances". Perfumer & Flavorist. 3 (1): 11–22.
- ↑ Naoki Mori; Kuwahara, Yasumasa; Kurosa, Kazuyoshi (1998). "Rosefuran: the sex pheromone of an acarid mite, Caloglyphus sp.". Journal of Chemical Ecology. 24 (11): 1771–1779. doi:10.1023/a:1022399331397.
- ↑ Büchi, George; Kovats, Erwin; Enggist, P.; Uhde, G . (1968). "Synthesis of rosefuran and dehydroelsholtzione". Journal of Organic Chemistry. 33 (3): 1227–9. doi:10.1021/jo01267a062.
- ↑ Seiichi Takano; Morimoto, Masamichi; Satoh, Shigeki (1984). "Syntheses of perillene and rosefuran from common starting materials". Chemistry Letters. 7: 1261–1262. doi:10.1246/cl.1984.1261.
- ↑ Ryozo Iriye; Uno, Tsuyoshi; Ohwa, Ikuo (1990). "The formation of alkylfurans from (E)-4-hydroxy-2-alkenals and (E)-4-oxo-2-alkenols, and a synthesis of rosefuran". Agricultural and Biological Chemistry. 54 (7): 1841–1843. doi:10.1271/bbb1961.54.1841.
- ↑ T. Bach; Krueger, L. (1998). "Regioselective Pd(0)-catalyzed coupling reactions on methyl 2,3-dibromofuran-5-carboxylate as a facile entry into 2,3,5-tri- and 2,3-disubstituted furans". Synlett. 11: 1185–1186. doi:10.1055/s-1998-1911.
- ↑ O. P. Vig; Vig, A. K; Handa, V. K. (1974). "Terpenoids. LXXXV. Claisen rearrangement involving the furan ring system. Synthesis of rosefuran and sesquirosefuran". Journal of the Indian Chemical Society. 51 (10): 900–902.
- ↑ Peter Weyerstahl; Schenk, Anja; Marschall, Helga (1995). "Structure-odor correlation. Part XXI. Olfactory properties and convenient synthesis of furans and thiophenes related to rosefuran and perillene and their isomers". Liebigs Annalen. 6 (10): 1849–1853. doi:10.1002/jlac.1995199510259.
- ↑ Hidetaka Tsukasa, "Manufacture of rosefuran", Jpn. Kokai Tokkyo Koho, 03151373 (1991)
- ↑ Gunther Ohloff (1978). "Importance of minor components in flavors and fragrances". Perfumer & Flavorist. 3 (1): 11–22.
- ↑ L. N. Misra; Husain, A. (1987). "The Essential Oil of Perilla ocimoides: A Rich Source of Rosefuran". Planta Medica. 53 (4): 379–380. doi:10.1055/s-2006-962743.
- ↑ rosefuran: http://www.thegoodscentscompany.com/data/rw1435551.html