Tetronic acid
 | |
| Names | |
|---|---|
| IUPAC name
4-Hydroxy-5H-furan-2-one | |
Other names
| |
| Identifiers | |
| 4971-56-6 (Keto form) 541-57-1 (Enol form) | |
| 3D model (Jmol) | Keto form: Interactive image Enol form: Interactive image |
| ChemSpider | 10301432 |
| ECHA InfoCard | 100.023.289 |
| EC Number | 208-785-3 |
| PubChem | 521261 |
| |
| Properties | |
| C4H4O3 | |
| Molar mass | 100.07 g·mol−1 |
| Melting point | 141–143 °C (286–289 °F; 414–416 K) (dec.)[1] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
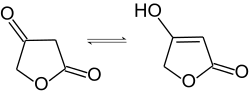
Tetronic acid is a chemical compound, classified as a γ-lactone, with the molecular formula C4H4O3.
It interconverts between keto and enol tautomers:[2]
Many natural products such as ascorbic acid (vitamin C), penicillic acid, pulvinic acids, and abyssomicins possess the β-keto-γ-butyrolactone motif of tetronic acid.[3]
In organic synthesis, it is used as a precursor for other substituted and ring-fused furans and butenolides.[4][5] It is also forms the structural core of a class of pesticides, known as tetronic acid insecticides, which includes spirodiclofen and spiromesifen.[6]
See also
References
- ↑ "2,4(3H,5H)-Furandione". Sigma-Aldrich.
- ↑ Abdou, Moaz M.; El-Saeed, Rasha A.; Abozeid, Mohamed A.; Elattar, Khaled M.; Zaki, E.G.; Barakat, Y.; Ibrahim, V.; Fathy, Mahmoud; Amine, M.; Bondock, Samir (2015). "Advancements in tetronic acid chemistry. Part 1: Synthesis and reactions". Arabian Journal of Chemistry. doi:10.1016/j.arabjc.2015.11.004.
- ↑ Georgiadis, Dimitris; Zografos, Alexandros (2006). "Synthetic Strategies towards Naturally Occurring Tetronic Acids". Synthesis. 2006 (19): 3157. doi:10.1055/s-2006-950202.
- ↑ "Tetronic acid". Alfa Aesar.
- ↑ Schmidt, Diane Grob; Seemuth, Paul D.; Zimmer, Hans (1983). "Substituted .gamma.-butyrolactones. Part 31. 2,4(3H,5H)-Furandione: Heteroannulations with aromatic o-amino carbonyl compounds and condensations with some vic-polyones". The Journal of Organic Chemistry. 48 (11): 1914. doi:10.1021/jo00159a029.
- ↑ "Classification of Insecticides". alanwood.net.
This article is issued from Wikipedia - version of the 8/24/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.