Titanium(II) oxide
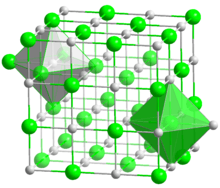 | |
| Names | |
|---|---|
| Other names
Titanium monoxide | |
| Identifiers | |
| 12137-20-1 | |
| 3D model (Jmol) | Interactive image |
| ECHA InfoCard | 100.032.020 |
| PubChem | 61685 |
| |
| |
| Properties | |
| TiO | |
| Molar mass | 63.866 g/mol |
| Appearance | bronze crystals |
| Density | 4.95 g/cm3 |
| Melting point | 1,750 °C (3,180 °F; 2,020 K) |
| Structure | |
| cubic | |
| Hazards | |
| Flash point | Non-flammable |
| Related compounds | |
| Titanium(III) oxide Titanium(III,IV) oxide Titanium(IV) oxide | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Titanium(II) oxide (TiO) is an inorganic chemical compound of titanium and oxygen. It can be prepared from titanium dioxide and titanium metal at 1500 °C.[1] It is non-stoichiometric in a range TiO0.7 to TiO1.3 and this is caused by vacancies of either Ti or O in the defect rock salt structure.[1] In pure TiO 15% of both Ti and O sites are vacant.[1] Careful annealing can cause ordering of the vacancies producing a monoclinic form which has 5 TiO units in the primitive cell that exhibits lower resistivity.[2] A high temperature form with titanium atoms with trigonal prismatic coordination is also known.[3] Acid solutions of TiO are stable for a short time then decompose to give hydrogen:[1]
- Ti2+ + H+ → Ti3+ + ½ H2
Evidence has been obtained for the presence of the diatomic molecule TiO in the interstellar medium.[4] TiO shows strong bands in the optical spectra of cool (M-type) stars.[5]
References
- 1 2 3 4 Holleman, A. F.; Wiberg, E. (2001), Inorganic Chemistry, San Diego: Academic Press, ISBN 0-12-352651-5
- ↑ Electrical and Magnetic Properties of TiO and VO, Banus M. D., Reed T. B., Strauss A. J., Phys. Rev. B 5, 2775 - 2784, (1972)doi:10.1103/PhysRevB.5.2775
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- ↑ Dyck, H. M.; Nordgren, Tyler E. "The effect of TiO absorption on optical and infrared angular diameters of cool stars" Astronomical Journal (2002), 124(1), 541-545. doi:10.1086/341039
- ↑ http://www.stsci.edu/~inr/ldwarf.html