Wanzlick equilibrium
The Wanzlick equilibrium is a chemical equilibrium between a relatively stable carbene compound and its dimer.
History
Original conjecture
In 1960, H.-W. Wanzlick and E. Schikora proposed that carbenes derived from dihydroimidazol-2-ylidene were generated by vacuum pyrolysis of 2-trichloromethyl dihydroimidazole derivatives, with the loss of chloroform.[1][2]
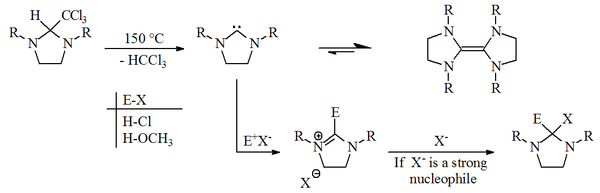
Wanzlick and Schikora believed that once prepared these carbenes existed in an unfavourable equilibrium with their corresponding dimers. This assertion was based on reactivity studies which they believed showed that the free carbene reacted with electrophiles (E-X). The dimer (a substituted tetraaminoethylene) was believed to be inactive to the electrophiles (E-X), and thought to merely act as a stable carbene reservoir.[3]
Conjecture challenged
Wanzlick’s hypothesis of a carbene-dimer equilibrium was tested by D. Lemal and others,[4] and separately by H. Winberg and others.[5] They heated together two tetraaminoethylene derivatives with different N-aryl substitututions:
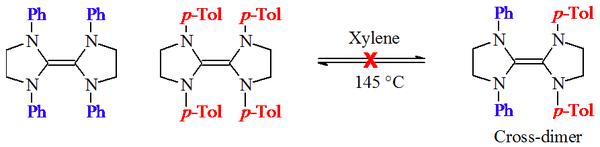
This reaction did not produce a mixed dimeric product, and accordingly indicated that a 'carbene-dimer equilibrium' did not exist for these dihydroimidazol-2-ylidene derivatives.
Lemal[4] proposed an alternative mechanism to account for the reactions observed by Wanzlick[3] by considering the reactivity of the electron rich tetraaminoethylenes — and not the stable carbenes, which at the time were still hypothetical.[2]
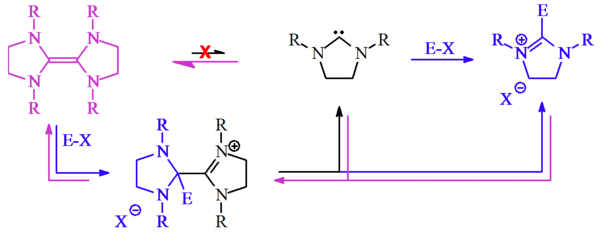
Lemal believed that the tetraaminoethylene, not the carbene, reacted with the electrophile (E-X) to generate a transient cationic species. He proposed that this cation then dissociated into the free carbene plus the resultant salt. The free carbene could then either re-dimerise (regenerating the tetraaminoethylene starting material) or react with E-X (as Wanzlick originally predicted), with either route eventually giving the same reaction product, the dihydroimidazolium salt.
Conjecture confirmed
In 1999 M. Denk reinvestigated the cross-over experiments and did find mixed dimers thus supporting the Wanzlick equilibrium.[7] This prompted Lemal to repeat his 1964 experiments. He was able to confirm Denk's findings but only with deuterated tetrahydrofuran (THF) as a solvent. With toluene and added KH as an electrophile quencher, however, the cross-over product was again not observed by Lemal.[8]
In 1999 Lemal [9] and Hahn [10] both investigated an equilibrium between a dibenzotetraazafulvalene derivative and its carbene, which led Böhm & Herrmann to conclude in 2000 that "the Wanzlick equilibrium between a tetraaminoethylene and its corresponding carbene did exist" after all.,[11] a notion confirmed in 2010 by Kirmse [12]
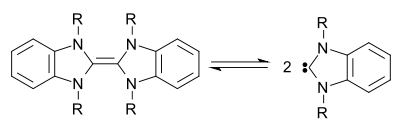
Further analysis
More recent work by Alder and others[13] has shown that unhindered diaminocarbenes form dimers by acid catalysed dimerisation as shown in the Lemal reaction above. In excess acid conditions the dimer forms the salt.
References
- ↑ Wanzlick Hans-Werner; Schikora E (1960). "Ein neuer Zugang zur Carben-Chemie". Angewandte Chemie. 72 (14): 494. doi:10.1002/ange.19600721409.
- 1 2 Wanzlick H. W.; Schikora E. (1960). "Ein nucleophiles Carben". Chemische Berichte. 94 (9): 2389–2393. doi:10.1002/cber.19610940905.
- 1 2 H. W. Wanzlick (1962). "Aspects of Nucleophilic Carbene Chemistry". Angew. Chem. Int. Ed. Engl. 1 (2): 75–80. doi:10.1002/anie.196200751.
- 1 2 D. M. Lemal; R. A. Lovald & K. I. Kawano (1964). "Tetraaminoethylenes. The Question of Dissociation". J. Am. Chem. Soc. 86 (12): 2518–2519. doi:10.1021/ja01066a044.
- ↑ H. E. Winberg; J. E. Carnahan; D. D. Coffman & M. Brown (1965). "Tetraaminoethylenes". J. Am. Chem. Soc. 87 (9): 2055–2056. doi:10.1021/ja01087a040.
- ↑ T. A. Taton & P. Chen (1996). "A Stable Tetraazafulvalene". Angew. Chem. Int. Ed. Engl. 35 (9): 1011–1013. doi:10.1002/anie.199610111.
- ↑ Denk Michael K.; Hatanoa Ken; Maa Martin (1999). "Nucleophilic carbenes and the wanzlick equilibrium: A reinvestigation". Tetrahedron Letters. 40 (11): 2057–2060. doi:10.1016/S0040-4039(99)00164-1.
- ↑ Liu Yufa; Lemal David M (2000). "Concerning the Wanzlick equilibrium". Tetrahedron Letters. 41 (5): 599–602. doi:10.1016/S0040-4039(99)02161-9.
- ↑ Liu Yufa; Lindner Patrick E.; Lemal David M. (1999). "Thermodynamics of a Diaminocarbene−Tetraaminoethylene Equilibrium". J. Am. Chem. Soc. 121 (45): 10626–10627. doi:10.1021/ja9922678.
- ↑ Hahn F. Ekkehardt; Wittenbecher Lars; Le Van Duc; Fröhlich Roland (2000). "Evidence for an Equilibrium between an N-heterocyclic Carbene and Its Dimer in Solution". Angewandte Chemie International Edition. 39 (3): 541–544. doi:10.1002/(sici)1521-3773(20000204)39:3<541::aid-anie541>3.0.co;2-b.
- ↑ Böhm Volker P. W.; Herrmann Wolfgang A. (2000). "The Wanzlick Equilibrium". Angewandte Chemie. 39 (22): 4036–4038. doi:10.1002/1521-3773(20001117)39:22<4036::AID-ANIE4036>3.0.CO;2-L.
- ↑ Kirmse W (2010). "The Beginnings of N-Heterocyclic Carbenes". Angewandte Chemie International Edition. 49 (47): 8798–8801. doi:10.1002/anie.201001658.
- ↑ Roger W. Alder; Leila Chaker; François P. V. Paolini (2004). "Bis(diethylamino)carbene and the mechanism of dimerisation for simple diaminocarbenes". Chem. Commun. (19): 2172–2173. doi:10.1039/b409112d. PMID 15467857.