2,5-Dimethylfuran
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2,5-Dimethylfuran | |
| Identifiers | |
| 625-86-5 | |
| 3D model (Jmol) | Interactive image Interactive image |
| ChemSpider | 11763 |
| ECHA InfoCard | 100.009.923 |
| EC Number | 210-914-3 |
| PubChem | 12266 |
| UNII | DR5HL9OJ7Y |
| |
| |
| Properties | |
| C6H8O | |
| Molar mass | 96.13 |
| Appearance | Liquid |
| Density | 0.8897 g/cm3 |
| Melting point | −62 °C (−80 °F; 211 K) |
| Boiling point | 92 to 94 °C (198 to 201 °F; 365 to 367 K) |
| Insoluble | |
| Refractive index (nD) |
1.44 - 1.442[1] |
| Hazards | |
| Main hazards | Very flammable, harmful |
| Flash point | −1 °C (30 °F; 272 K) [1] |
| 285.85[2] | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
2,5-Dimethylfuran is a heterocyclic compound with the formula (CH3)2C4H2O. Although often abbreviated DMF, it should not be confused with dimethylformamide. A derivative of furan, this simple compound is a potential biofuel, being derivable from cellulose.
Production
Fructose can be converted into 2,5-dimethylfuran in a catalytic biomass-to-liquid process. The conversion of fructose to DMF proceeds via hydroxymethylfurfural.[3][4]
Fructose is obtainable from glucose, a building block in cellulose.[5][6]
Potential as a biofuel
DMF has a number of attractions as a biofuel. It has an energy density 40% greater than that of ethanol, making it comparable to gasoline (petrol). It is also chemically stable and, being insoluble in water, does not absorb moisture from the atmosphere. Evaporating dimethylfuran during the production process also requires around one third less energy than the evaporation of ethanol,[3][7] although it has a boiling point some 14 °C higher, at 92 °C, compared to 78 °C for ethanol.
The ability to efficiently and rapidly produce dimethylfuran from fructose, found in fruit and some root vegetables, or from glucose, which can be derived from starch and cellulose - all widely available in nature - adds to the attraction of dimethylfuran, although safety issues must be examined. Bioethanol and biodiesel are currently the leading liquid biofuels.
The stoichiometric air/fuel ratio of dimethylfuran is 10.72, compared to ethanol at 8.95 and gasoline at 14.56.[2] This means that burning dimethylfuran requires approximately 33% less air than the same quantity of gasoline, but approximately 20% more air than the same quantity of ethanol.
The calorific value of liquid dimethylfuran is 33.7 MJ/kg, compared to 26.9 MJ/kg for ethanol and 43.2 MJ/kg for gasoline.[2] The research octane number (RON) of dimethylfuran is 119.[2] The latent heat of vaporization at 20 °C is 31.91 kJ/mol.[2] Recent tests in a single-cylinder gasoline engine found that the thermal efficiency of burning dimethylfuran is similar to that of gasoline.[8]
Other uses
2,5-Dimethylfuran serves as a scavenger for singlet oxygen, a property which has been exploited for the determination of singlet oxygen in natural waters. The mechanism involves a Diels-Alder reaction followed by hydrolysis, ultimately leading to diacetylethylene and hydrogen peroxide as products. More recently, furfuryl alcohol has been used for the same purpose.[9]
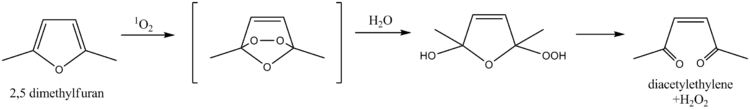
This compound has also been proposed as an internal standard for NMR spectroscopy. 2,5-Dimethylfuran has singlets in its 1H NMR spectrum at δ 2.2 and 5.8; the singlets give reliable integrations, while the positions of the peaks do not interfere with many analytes. The compound also has an appropriate boiling point of 92 °C which prevents evaporative losses, yet is easily removed.[10]
Role in food chemistry
2,5-Dimethylfuran forms upon thermal degradation of some sugars and has been identified in trace amounts as a component of caramelized sugars.[11]
Toxicology
2,5-Dimethylfuran plays a role in the mechanism for the neurotoxicity of hexane in humans. Together with hexane-2,5-dione and 4,5-dihydroxy-2-hexanone, it is one of the main metabolites of hexane.[12]
2,5-Dimethylfuran has been identified as one of the components of cigar smoke with low cilatoxicity (ability to adversely affect the cilia in the respiratory tract that are responsible for removing foreign particles).[13] Its blood concentration can be used as a biomarker for smoking.[14]
Comparison of MSDS sheets shows that human handling of 2,5-dimethylfuran[15][16][17] is approximately as hazardous as handling gasoline.[18][19][20]
References
- 1 2 "ChemExper chemical directory - Catalog of chemicals and suppliers".
- 1 2 3 4 5 "Direct-Injection Engine Study Finds That DMF Is a Promising Biofuel, With Combustion Performance and Regulated Emissions Comparable to Gasoline | Eco Friendly".
- 1 2 Yuriy Román-Leshkov; Christopher J. Barrett; Zhen Y. Liu; James A. Dumesic (2007). "Production of dimethylfuran for liquid fuels from biomass-derived carbohydrates". Nature. 447 (7147): 982–5. doi:10.1038/nature05923. PMID 17581580.
- ↑ Matt McGrath (2007-06-21). "Fruit could make 'powerful fuel'". BBC News. Retrieved 2007-06-22.
- ↑ Haibo Zhao; Johnathan E. Holladay; Heather Brown; Z. Conrad Zhang (June 15, 2007). "Metal Chlorides in Ionic Liquid Solvents Convert Sugars to 5-Hydroxymethylfurfural". Science. 316 (5831): 1597–1600. doi:10.1126/science.1141199. PMID 17569858.
- ↑ "Plastic that grows on trees". Pacific Northwest National Laboratory. 2007-06-21. Archived from the original (press release) on 13 July 2007. Retrieved 2007-06-22.
- ↑ James Beal (2007-06-20). "Engineers develop higher-energy liquid-transportation fuel from sugar" (press release). University of Wisconsin-Madison. Archived from the original on 1 July 2007. Retrieved 2007-06-22.
- ↑ "Green Car Congress: Direct-Injection Engine Study Finds That DMF Is a Promising Biofuel, With Combustion Performance and Regulated Emissions Comparable to Gasoline".
- ↑ Patrick L. Brezonik. Chemical Kinetics and Process Dynamics in Aquatic Systems. CRC Press, 1994, p. 671.
- ↑ S. W. Gerritz; A. M. Sefler (2000). "2,5-Dimethylfuran (DMFu): An Internal Standard for the "Traceless" Quantitation of Unknown Samples via 1H NMR". J. Comb. Chem. 2 (1): 39–41. doi:10.1021/cc990041v.
- ↑ W.D. Powrie; C.H. Wu; V.P. Molund (1986). "Browning reaction systems as sources of mutagens and antimutagens". Environmental Health Perspectives. 67: 47–54. doi:10.2307/3430317. JSTOR 3430317. PMC 1474410
 . PMID 3757959.
. PMID 3757959. - ↑ Peter Arlien-Søborg. Solvent Neurotoxicity. CRC Press, 1992. ISBN 0-8493-6234-2.
- ↑ Donald Shopland. Cigars: Health Effects and Trends. DIANE Publishing, 1998.
- ↑ Ashley, D. L.; Bonin, M. A.; Hamar, B.; McGeehin, M. (1996). "Using the blood concentration of 2,5-dimethylfuran as a marker for smoking". International Archives of Occupational and Environmental Health. 68 (3): 183–7. doi:10.1007/BF00381629. PMID 8919847.
- ↑ "fishersci.com".
- ↑ "chemexper.com".
- ↑ "pennakem.com" (PDF).
- ↑ "Equiva Services - MSDS".
- ↑ "hazard.com".
- ↑ "hazard.com".