Glucocerebrosidase
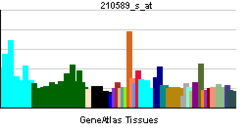
| View/Edit Human | View/Edit Mouse |
β-Glucocerebrosidase (also called acid β-glucosidase, D-glucosyl-N-acylsphingosine glucohydrolase, or GCase) is an enzyme with glucosylceramidase activity (EC 3.2.1.45) that is needed to cleave, by hydrolysis, the beta-glucosidic linkage of the chemical glucocerebroside, an intermediate in glycolipid metabolism. It is localized in the lysosome and has a molecular weight of 59700 Daltons.
Clinical significance
Mutations in the glucocerebrosidase gene cause Gaucher's disease, a lysosomal storage disease characterized by an accumulation of glucocerebrosides. A related pseudogene is approximately 12 kb downstream of this gene on chromosome 1. Alternative splicing results in multiple transcript variants encoding the same protein.[3]
Mutations in the glucocerebrosidase gene are also associated with Parkinson's disease.[4][5]
Drugs
Alglucerase (Ceredase) was a version of glucocerebrosidase that was harvested from human placental tissue and then modified with enzymes.[6] It was approved by the FDA in 1991[7] and has been withdrawn from the market[8][9] due to the approval of similar drugs made with recombinant DNA technology instead of being harvested from tissue; drugs made recombinantly, since there is no concern about diseases being transmitted from the tissue used in harvesting, and are less expensive to manufacture.[6]
Recombinant glucocerebrosidases used as drugs include:[10]
- Imiglucerase (Cerezyme) [6]
- Velaglucerase (Vpriv) [6]
- Taliglucerase alfa (Elelyso) [11]
See also
- Closely related enzymes
References
- ↑ "Human PubMed Reference:".
- ↑ "Mouse PubMed Reference:".
- ↑ "Entrez Gene: GBA glucosidase, beta; acid (includes glucosylceramidase)".
- ↑ "PDGene". Alzheimer Research Forum. 2008-11-12. Retrieved 2008-11-13.
- ↑ Lwin A, Orvisky E, Goker-Alpan O, LaMarca ME, Sidransky E (January 2004). "Glucocerebrosidase mutations in subjects with parkinsonism". Molecular Genetics and Metabolism. 81 (1): 70–3. doi:10.1016/j.ymgme.2003.11.004. PMID 14728994.
- 1 2 3 4 Deegan PB, Cox TM (2012). "Imiglucerase in the treatment of Gaucher disease: a history and perspective". Drug Design, Development and Therapy. 6: 81–106. doi:10.2147/DDDT.S14395. PMC 3340106
 . PMID 22563238.
. PMID 22563238. - ↑ "Regulatory Matters" (PDF). WHO Drug Information. 5 (3): 123–4. 1991.
- ↑ "Enzyme-replacement Therapy for Lysosomal Storage Disorders". Clinical Policy Bulletin Number: 0442. Aetna. 2014-08-08.
- ↑ "FDA Prescription and Over-the-Counter Drug Product List" (PDF). Additions/Deletions for Prescription Drug Product List. U.S. Food and Drug Administration. March 2012.
- ↑ Grabowski GA (2012). "Gaucher disease and other storage disorders". Hematology / the Education Program of the American Society of Hematology. American Society of Hematology. Education Program. 2012: 13–8. doi:10.1182/asheducation-2012.1.13. PMID 23233555.
- ↑ Yukhananov, Anna (1 May 2012). "U.S. FDA approves Pfizer/Protalix drug for Gaucher". Chicago Tribune. Reuters. Retrieved 2 May 2012.
Further reading
- Horowitz M, Zimran A (1994). "Mutations causing Gaucher disease". Human Mutation. 3 (1): 1–11. doi:10.1002/humu.1380030102. PMID 8118460.
- Tayebi N, Stone DL, Sidransky E (October 1999). "Type 2 gaucher disease: an expanding phenotype". Molecular Genetics and Metabolism. 68 (2): 209–19. doi:10.1006/mgme.1999.2918. PMID 10527671.
- Stone DL, Tayebi N, Orvisky E, Stubblefield B, Madike V, Sidransky E (2000). "Glucocerebrosidase gene mutations in patients with type 2 Gaucher disease". Human Mutation. 15 (2): 181–8. doi:10.1002/(SICI)1098-1004(200002)15:2<181::AID-HUMU7>3.0.CO;2-S. PMID 10649495.
- Caillaud C, Poenaru L (2002). "[Gaucher's and Fabry's diseases: biochemical and genetic aspects]". Journal De La Société De Biologie. 196 (2): 135–40. PMID 12360742.
- Fabrega S, Durand P, Mornon JP, Lehn P (2002). "[The active site of human glucocerebrosidase: structural predictions and experimental validations]". Journal De La Société De Biologie. 196 (2): 151–60. PMID 12360744.
- Alfonso P, Aznarez S, Giralt M, Pocovi M, Giraldo P (2007). "Mutation analysis and genotype/phenotype relationships of Gaucher disease patients in Spain". Journal of Human Genetics. 52 (5): 391–6. doi:10.1007/s10038-007-0135-4. PMID 17427031.
External links
- GeneReviews/NCBI/UW/NIH entry on Gaucher disease
- Glucocerebrosidase at the US National Library of Medicine Medical Subject Headings (MeSH)
- Proteopedia Acid-beta-glucosidase