Nuclear RNase P
| Nuclear RNase P | |
|---|---|
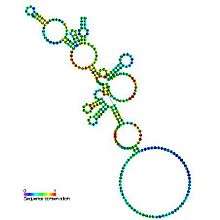 | |
| Predicted secondary structure and sequence conservation of RNaseP_nuc | |
| Identifiers | |
| Symbol | RNaseP_nuc |
| Rfam | RF00009 |
| Other data | |
| RNA type | Gene; ribozyme |
| Domain(s) | Eukaryota; Bacteria; Archaea |
| GO | 0008033 0004526 0030677 |
| SO | 0000386 |
In molecular biology, nuclear ribonuclease P (RNase P) is a ubiquitous endoribonuclease, found in archaea, bacteria and eukarya as well as chloroplasts and mitochondria. Its best characterised enzyme activity is the generation of mature 5'-ends of tRNAs by cleaving the 5'-leader elements of precursor-tRNAs. Cellular RNase Ps are ribonucleoproteins. The RNA from bacterial RNase P retains its catalytic activity in the absence of the protein subunit, i.e. it is a ribozyme. Similarly, archaeal RNase P RNA has been shown to be weakly catalytically active in the absence of its respective protein cofactors.[1] Isolated eukaryotic RNase P RNA has not been shown to retain its catalytic function, but is still essential for the catalytic activity of the holoenzyme. Although the archaeal and eukaryotic holoenzymes have a much greater protein content than the bacterial ones, the RNA cores from all three lineages are homologous -- the helices corresponding to P1, P2, P3, P4, and P10/11 are common to all cellular RNase P RNAs. Yet there is considerable sequence variation, particularly among the eukaryotic RNAs.
References
- ↑ Tsai, HY; Pulukkunat, DK; Woznick, WK; Gopalan V (2006). "Functional reconstitution and characterization of Pyrococcus furiosus RNase P". PNAS 103: 16147-16152.
Further reading
- Frank, DN; Pace NR (1998). "Ribonuclease P: unity and diversity in a tRNA processing ribozyme". Annu Rev Biochem. 67: 153–180. doi:10.1146/annurev.biochem.67.1.153. PMID 9759486.
- Brown, JW (1999). "The Ribonuclease P Database". Nucleic Acids Res. 27 (1): 314–. doi:10.1093/nar/27.1.314. PMC 148169
 . PMID 9847214.
. PMID 9847214. - Frank, DN; Adamidi C; Ehringer MA; Pitulle C; Pace NR (2000). "Phylogenetic-comparative analysis of the eukaryal ribonuclease P RNA". RNA. 6 (12): 1895–1904. doi:10.1017/S1355838200001461. PMC 1370057
 . PMID 11142387.
. PMID 11142387. - Xiao, S; Scott F; Fierke CA; Engelke DR (2002). "Eukaryotic Ribonuclease P: A Plurality of Ribonucleoprotein Enzymes". Annu Rev Biochem. 71: 165–189. doi:10.1146/annurev.biochem.71.110601.135352. PMID 12045094.
- Marquez, SM; Harris JK; Kelley ST; Brown JW; Dawson SC; Roberts EC; Pace NR (2005). "Structural implications of novel diversity in eucaryal RNase P RNA". RNA. 11 (5): 739–751. doi:10.1261/rna.7211705. PMC 1370759
 . PMID 15811915.
. PMID 15811915.