Mycoplasma laboratorium
Mycoplasma laboratorium is a designed, partially synthetic species of bacterium derived from the genome of Mycoplasma genitalium. This effort in synthetic biology is being undertaken at the J. Craig Venter Institute by a team of approximately 20 scientists headed by Nobel laureate Hamilton Smith, and including DNA researcher Craig Venter and microbiologist Clyde A. Hutchison III. Mycoplasma genitalium was chosen as it was the species with the smallest number of genes known at that time.
On May 21, 2010, Science reported that the Venter group had successfully synthesized the genome of the bacterium Mycoplasma mycoides from a computer record, and transplanted the synthesized genome into the existing cell of a Mycoplasma capricolum bacterium that had had its DNA removed. The "synthetic" bacterium was viable, i.e. capable of replicating billions of times. (The team had originally planned to use the M. genitalium bacterium they had previously been working with, but switched to M. mycoides because the latter bacterium grows much faster, which translated into quicker experiments.) Scientists who were not involved in the study caution that it is not a truly synthetic life form because its genome was put into an existing cell.[b 1]
It is estimated that the synthetic genome cost US$40 million to make and took 20 people more than a decade of work.[b 2] Despite the controversy, Venter has attracted over $110 million in investments so far for Synthetic Genomics, with a future deal with Exxon Mobil of $300 million in research to design algae for diesel fuel.[b 3]
Mycoplasma
Mycoplasma genitalium was chosen as it was the species with the smallest number of genes known at that time.
Mycoplasma is a genus of bacteria of the class Mollicutes in the division Tenericutes, characterised by the lack of a cell wall (making it Gram negative) due to their parasitic or commensal lifestyle (extracellular and intracellular). In molecular biology, the genus has received much attention. Apart from being a notorious and hard to eradicate (immune to beta-lactam and other antibiotics) contaminant in mammalian cell cultures,[a 1] it has also been used as a model organism: the second published complete bacterial genome sequence was that of Mycoplasma genitalium, which has one of the smallest genomes of free-living organisms.[a 2] The M. pneumoniae genome sequence was published soon afterward and was the first genome sequence determined by primer walking of a cosmid library instead of the whole-genome shotgun method.[a 3] Consequently, this species was chosen as a model for the minimal cell project,[a 4] catalog the entire protein content of a cell.[a 5]
Other genera
Pelagibacter ubique (an α-proteobacterium of the order Rickettsiales) has the smallest known genome (1,308,759 base pairs) of any free living organism and is one of the smallest self-replicating cells known. It is possibly the most numerous bacterium in the world (perhaps 1028 individual cells) and, along with other members of the SAR11 clade, are estimated to make up between a quarter and a half of all bacterial or archaeal cells in the ocean.[a 6] However, this species was identified only in 2002 by rRNA sequences and was fully sequenced in 2005,[a 7] being an extremely hard to cultivate species which does not reach a high growth density,[a 8][a 9] Additionally, several newly discovered species have fewer genes than M. genitalium, but many essential genes that are missing in Hodgkinia cicadicola, Sulcia muelleri, Baumannia cicadellinicola (symbionts of cicadas) and Carsonella ruddi (symbiote of hackberry petiole gall psyllid, Pachypsylla venusta[a 10]) may be encoded in the host nucleus as these endosymbionts are acquiring an organelle-like status in a similar way to mitochondria and chloroplasts.[a 11]
Minimal genome project
The team started with the bacterium M. genitalium, an obligate intracellular parasite whose genome consists of 482 genes comprising 582,970 base pairs, arranged on one circular chromosome (the smallest genome of any known natural organism that can be grown in free culture). They then systematically removed genes to find a minimal set of 382 genes that can sustain life.[a 12] This effort was also known as the Minimal Genome Project.
The team intends to synthesize chromosome DNA sequences consisting of these 382 genes. Once a version of the minimal 382-gene chromosome has been synthesized, it is intended to be transplanted into a M. genitalium cell to create Mycoplasma laboratorium.
The resulting Mycoplasma laboratorium bacterium is expected to be able to replicate itself with its man-made DNA, making it the most synthetic organism to date, although the molecular machinery and chemical environment that would allow it to replicate would not be synthetic.[b 4]
In December 2003, the team had reported a fast method of synthesizing a genome from scratch, producing the 5386-base genome of the bacteriophage Phi X 174 in about two weeks.[a 13] However, the genome of Mycoplasma laboratorium is about 50 times larger. In January 2008, the team reported to have synthesized the complete 582,970 base pair chromosome of M. genitalium, with small modifications so that it won't be infectious and can be distinguished from the wild type. They named this genome Mycoplasma genitalium JCVI-1.0.[a 14][b 5] The team had also demonstrated the process of transplanting a (non-synthetic) genome from one Mycoplasma species to another in June 2007.[b 6] In May 2010 they showed that they were able to synthesize the 1,078,809 base pair genome[a 15] of Mycoplasma mycoides from scratch and transplant it into a Mycoplasma capricolum cell; the new genome then took over the cell and the new organism multiplied.[b 7] The new organism was nicknamed Synthia.
Venter hopes to eventually synthesize bacteria to manufacture hydrogen and biofuels, and also to absorb carbon dioxide and other greenhouse gases. George M. Church, another pioneer in synthetic biology, holds that E. coli is a more efficient organism than M. genitalium and that creating a fully synthetic genome is not necessary and too costly for such tasks; he points out that synthetic genes have already been incorporated into E.coli to perform some of the above tasks.[b 5] On June 28, 2007, a team at the J. Craig Venter Institute published an article in Science Express, saying that they had successfully transplanted the natural DNA from a Mycoplasma mycoides bacterium into a Mycoplasma capricolum cell, creating a bacterium which behaved like a M. mycoides.[a 16]
On Oct 6, 2007, Craig Venter announced in an interview with UK's The Guardian newspaper that the same team had synthesized a modified version of the single chromosome of Mycoplasma genitalium using chemicals. The chromosome was modified to eliminate all genes which tests in live bacteria had shown to be unnecessary. The next planned step in this minimal genome project is to transplant the synthesized minimal genome into a bacterial cell with its old DNA removed; the resulting bacterium will be called Mycoplasma laboratorium. The next day the Canadian bioethics group, ETC Group issued a statement through their representative, Pat Mooney, saying Venter's "creation" was "a chassis on which you could build almost anything". The synthesized genome had not yet been transplanted into a working cell.[b 4]
On May 21, 2010, Science reported that the Venter group had successfully synthesized the genome of the bacterium Mycoplasma mycoides from a computer record, and transplanted the synthesized genome into the existing cell of a Mycoplasma capricolum bacterium that had had its DNA removed. The "synthetic" bacterium was viable, i.e. capable of replicating billions of times. The team had originally planned to use the M. genitalium bacterium they had previously been working with, but switched to M. mycoides because the latter bacterium grows much faster, which translated into quicker experiments.[b 2] They have also shown that the natural genome of M. mycoides can be transplanted but has yet to show that the same could be done for M. genitalium.[b 8] Venter describes it as "the first species.... to have its parents be a computer".[b 9] The transformed bacterium is dubbed "Synthia" by ETC. A Venter spokesperson has declined to confirm any breakthrough at the time of this writing, likely because similar genetic introduction techniques such as transfection, transformation, transduction and protofection have been a standard research practice for many years.
Now that the technique has been proven to work with the M. mycoides genome, the next project is presumably to go back to the minimized M. genitalium and transplant it into a cell to create the previously mentioned Mycoplasma laboratorium.
The creation of a new synthetic bacterium was announced in Science on March 25, 2016. It has only 473 genes, the fewest genes of any freely living organism. This fast growing new cell, called Syn 3.0, was created by transplanting the genome of Mycoplasma mycoides, with all the unessential DNA removed, into a Mycoplasma capricolum, which had been emptied of its own DNA.
M. genitalium was not used because it reproduces too slowly.
Bacterial genome transplantation
In order to propagate a synthetic genome, the technique to transplant an intact whole bacterial genome into another had to be developed. Oswald Avery's pioneering experiments in the 1940s showed that some bacteria could take up naked DNA,[a 17] and with the advent of molecular cloning techniques DNA elements could be transformed into competent cells, typically cloning vectors, around 5-20 kbp long, and even bacterial artificial chromosomes can be maintained. In 2007, Venter's team reported that they had managed to transfer the chromosome of the species Mycoplasma mycoides to Mycoplasma capricolum by means of:
- isolating the genome of M. mycoides: gentle lysis of cells trapped in agar—molten agar mixed with cells and left to form a gel—followed by pulse field gel electrophoresis and the band of the correct size (circular 1.25Mbp) being isolated;
- making the recipient cells of M. capricolum competent: growth in rich media followed starvation in poor media where the nucleotide starvation results in inhibition of DNA replication and change of morphology; and
- polyethylene glycol-mediated transformation of the circular chromosome to the DNA-free cells followed by selection.[a 16]
The term transformation is used to refer to insertion of a vector into a bacterial cell (by electroporation or heatshock). Here, transplantation is used akin to nuclear transplantation.
The switch from M. genitalium to M. mycoides was spurred due to the faster growth of the latter.[b 2]
Bacterial chromosome synthesis
It is possible to create DNA sequences chemically (oligonucleotide synthesis) which is achieved by successive rounds of deprotection and coupling of protected phosphoramidite nucleotides with geometrically decreasing yields to length, making sequences longer than 1kb unfeasible. For longer sequences, DNA ligation is required. In 2008 Venter's group published a paper showing that they had managed to create a synthetic genome (a copy of M. mycoides sequence CP001621) by means of a hierarchical strategy:[a 14]
- Synthesis → 1kbp: The genome sequence was synthesized by Blue Heron in 1078 1080bp cassettes with 80bp overlap and NotI restriction sites (inefficient but rare cutter).
- Ligation → 10kbp: 109 Groups of a series of 10 consecutive cassettes were ligated and cloned in E.coli on a plasmid and the correct permutation checked by sequencing, this would follow a geometric distribution with expected number of trials of 10.
- Multiplex PCR → 100kbp: 11 Groups of a series of 10 consecutive 10kbp assemblies (grown in yeast) were joined by multiplex PCR, using a primer pair for each 10kbp assembly.
- Isolation and recombination → secondary assemblies were isolated by means of the plug method above and joining and transformed into yeast spheroplasts without a vector sequence (present in assembly 811-900).
Synthetic genome
In 2010, using the methods described above, Venter and colleagues created a strain of Mycoplasma mycoides called JCVI-syn1.0 with a synthetic genome.[a 18] Initially the synthetic construct did not work, so to pin point the error—which caused a delay of 3 months in the whole project[b 2]—a series of semi-synthetic constructs were created. Given the fact that the natural genome worked, the cause of the failed growth was an frameshift mutation in DnaA, a replication initiation factor, which, once corrected, worked and was verified.[a 18]
The construction of a cell with a synthetic genome was done to test the methodology—allowing more modified genomes to be created in the future. To minimize sources of failure, the genome was created using a natural genome as a template. Due to the large size of a genome, apart from the elements required for propagation in yeast and residues from restriction sites, several differences are present in Mycoplasma mycoides JCVI-syn1.0 notably an E.coli transposon IS1 (an infection from the 10kb stage) and an 85bp duplication.[a 18]
However, the project has received heavy criticism as it claims to have created a synthetic organism. This claim arises from the fact that the genome was synthesized chemically in many pieces (a synthetic method), joined together by means of molecular biological techniques (an artificial method), and transplanted into the cytoplasm of a natural cell (after a few generations, though, the original protein content is undetectable). The two species used as donor and recipient are of the same genus as the more distant two species are, the less the correct protein interactions are maintained, such as binding factors and binding sites, which mutate together (epistasis).[a 19] Consequently, Paul Keim (a molecular geneticist at Northern Arizona University in Flagstaff) notes that "there are great challenges ahead before genetic engineers can mix match, and fully design an organism's genome from scratch" [b 2] due to this issue. DNA is the template for protein construction and requires proteins as helper molecules to do so, a chicken-and-egg conundrum solved by the RNA world hypothesis, consequently synthetic naked DNA would require several proteins to create a viable cell. In 2000 and 2002 teams synthesized replicating hepatitis C virus (about 9600 nucleotides long)[a 20] and poliovirus (about 7500 nucleotides long),[a 21] Viruses, however, replicate by utilising host protein expression machinery. Furthermore, whereas DNA can easily be replicated (using DNA polymerase), transcribed (using RNA polymerase), and translated (using ribosomes and many other factors)—all in vitro, such reactions, so far, utilise cell extracts and most components have not been synthesized de novo—that is from inorganic or synthetically made organic chemicals only.
Watermarks

A much publicised feature of the Mycoplasma laboratorium is the presence of watermark sequences as an ultimate proof of the achievement and as a publicity stunt—similar to the tradition of chip art, inscriptions on unused portions of microchips visible only by electron microscopy. The 4 watermarks (present in figure 1 in supplementary material of [a 18]) are coded messages in the form of DNA base pairs, of 1246, 1081, 1109 and 1222 base pairs respectively, in natural peptides the 4 nucleotides encode in sets of 3 the 20 natural amino acids by means of the standard genetic code. Each amino acid by convention is represented by a letter, but in nature there is nothing which ties Alanine, a molecule, to the Latin letter A, a vowel, so this convention was disregarded in the latter watermarks. In the minimal genome organism the watermark were encoded as amino acids, with V as U, both in reference to Latin inscriptions and the lack of a standard amino acid for U containing the names of the researchers: (VENTERINSTITVTE, CRAIGVENTER, HAMSMITH, CINDIANDCLYDE, GLASSANDCLYDE). In the synthetic organism, instead the Latin alphabet—which in English has 26 letters, which is covered only in base 4 with 3 or more digits—was encoded by an undisclosed encoding. The encoding is fixed and 3 digits make an uppercase letter or Ascii symbol, possibly randomly allocated (not Ascii table, frequency or keyboard order).[b 10] The content of the watermarks is as follows:
- watermark 1 an Html script which reads to a browser as text congratulating the decoder with an email link to click to prove the decoding.
- watermark 2 contains a list of authors and a quote from James Joyce: "To live, to err, to fall, to triumph, to recreate life out of life".
- watermark 3 contains more authors and a quote from Robert Oppenheimer (uncredited): "See things not as they are, but as they might be".
- watermark 4 contains yet more authors and a quote from Richard Feynman: "What I cannot build, I cannot understand".
Concerns and controversy
Press coverage
The main controversy from the project is the undue amount of publicity it received from the press due to Venter's showmanship, to the degree that Jay Keasling, a pioneering synthetic biologist and founder of Amyris says "The only regulation we need is of my colleague's mouth".[b 3]
Utility
Despite the funding for practical applications, as stressed by George M. Church, one of the main players in the field of synthetic biology, a few changes are required to obtain useful organisms now, such as biofuel production or bioremediation.[b 3] However, speculation about the distant future's possible applications is rife. Venter himself is prone to such speculations, having asked for example, "What if we can make algae taste like beef?"[b 3] If it were possible to create a synthetic cell without the use of preexisting recipient cells, however, many applications would become achievable which would be otherwise unattainable, such as a completely overhauled bacterium that works in a logically controlled way—removing what has been described as 'evolutionary messiness'[a 22]—with lower mutation rates, categorical gene arrangement (colinearity), the possibility of adding novel nucleotides to increase encoding (a feat achieved in vitro (PCR)); or with a completely novel genetic code, such as has been achieved by experiments in which a few additional non-canonical amino acids were added.
Intellectual property
The J. Craig Venter Institute filed patents for the Mycoplasma laboratorium genome (the "minimal bacterial genome") in the U.S. and internationally in 2006.[b 11][b 12][a 23] This extension of the domain of biological patents is being challenged by the watchdog organization Action Group on Erosion, Technology and Concentration.[b 13]
JCVI-syn3.0
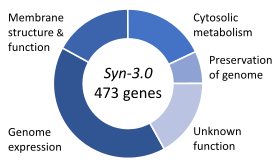
In 2016, the Venter Institute used genes from JCVI-syn1.0 to synthesize an even smaller genome they call JCVI-syn3.0, that contains 531,560 base pairs and 473 genes.[2] Originally in 1996, after comparing M. genitalium with another small bacterium Haemophilus influenza, Arcady Mushegian and Eugene Koonin had proposed that there might be a common set of 256 genes which could be a minimal set of genes needed for viability.[3][4] In this new organism, the number of genes can only be pared down to 473, 149 of which whose functions are completely unknown.[3] The organism with the smallest known set of genes is Nasuia deltocephalinicola which has only 137 genes,[5] but the minimal genome for any free-living organism would be different depending on its environment.[3]
Similar projects
From 2002 to 2010, a team at the Hungarian Academy of Science created a strain of Escherichia coli called MDS42, which is now sold by Scarab Genomics of Madison, WI under the name of "Clean Genome. E.coli",[b 14] where 15% of the genome of the parental strain (E. coli K-12 MG1655 ) were removed to aid in molecular biology efficiency, removing IS elements, pseudogenes and phages, resulting in better maintenance of plasmid-encoded toxic genes, which are often inactivated by transposons.[a 24][a 25][a 26] Biochemistry and replication machinery were not altered.
References
Primary sources
- ↑ Young L, Sung J, Stacey G, Masters JR. "Detection of Mycoplasma in cell cultures". Nat Protoc. 2010 5(5): 929–34. Epub 2010 Apr 22.
- ↑ Fraser CM, Gocayne JD, White O, et al. (October 1995). "The minimal gene complement of Mycoplasma genitalium". Science. 270 (5235): 397–403. Bibcode:1995Sci...270..397F. doi:10.1126/science.270.5235.397. PMID 7569993.
- ↑ Himmelreich R, Hilbert H, Plagens H, Pirkl E, Li BC, Herrmann R (November 1996). "Complete sequence analysis of the genome of the bacterium Mycoplasma pneumoniae". Nucleic Acids Research. 24 (22): 4420–49. doi:10.1093/nar/24.22.4420. PMC 146264
 . PMID 8948633.
. PMID 8948633. - ↑ Hutchison CA, Montague MG (2002). "Mycoplasmas and the minimal genome concept". Molecular Biology and Pathogenicity of Mycoplasmas (Razin S, Herrmann R, eds.). New York: Kluwer Academic/Plenum. pp. 221–54. ISBN 0-306-47287-2.
- ↑ Regula JT, Ueberle B, Boguth G, et al. (November 2000). "Towards a two-dimensional proteome map of Mycoplasma pneumoniae". Electrophoresis. 21 (17): 3765–80. doi:10.1002/1522-2683(200011)21:17<3765::AID-ELPS3765>3.0.CO;2-6. PMID 11271496.
- ↑ Morris RM, et al. (2002). "SAR11 clade dominates ocean surface bacterioplankton communities". Nature. 420 (6917): 806–10. Bibcode:2002Natur.420..806M. doi:10.1038/nature01240. PMID 12490947.
- ↑ Stephen J. Giovannoni, H. James Tripp, et al. (2005). "Genome Streamlining in a Cosmopolitan Oceanic Bacterium". Science. 309 (5738): 1242–1245. Bibcode:2005Sci...309.1242G. doi:10.1126/science.1114057. PMID 16109880.
- ↑ Rappé MS, Connon SA, Vergin KL, Giovannoni SL (2002). "Cultivation of the ubiquitous SAR11 marine bacterioplankton clade". Nature. 418 (6898): 630–33. Bibcode:2002Natur.418..630R. doi:10.1038/nature00917. PMID 12167859.
- ↑ Tripp HJ, Kitner JB, Schwalbach MS, Dacey JW, Wilhelm LJ, Giovannoni SJ (Apr 10, 2008). "SAR11 marine bacteria require exogenous reduced sulphur for growth". Nature. 452 (7188): 741–4. Bibcode:2008Natur.452..741T. doi:10.1038/nature06776. PMID 18337719.
- ↑ Nakabachi, A.; Yamashita, A.; Toh, H.; Ishikawa, H.; Dunbar, H. E.; Moran, N. A.; Hattori, M. (2006). "The 160-Kilobase Genome of the Bacterial Endosymbiont Carsonella". Science. 314 (5797): 267. doi:10.1126/science.1134196. PMID 17038615.
- ↑ McCutcheon, J. P.; McDonald, B. R.; Moran, N. A. (2009). "Convergent evolution of metabolic roles in bacterial co-symbionts of insects". Proceedings of the National Academy of Sciences. 106 (36): 15394–15399. Bibcode:2009PNAS..10615394M. doi:10.1073/pnas.0906424106. PMC 2741262
 . PMID 19706397.
. PMID 19706397. - ↑ Glass, John I.; Nacyra Assad-Garcia; Nina Alperovich; Shibu Yooseph; Matthew R. Lewis; Mahir Maruf; Clyde A. Hutchison; Hamilton O. Smith; J. Craig Venter (2006-01-10). "Essential genes of a minimal bacterium". Proceedings of the National Academy of Sciences. 103 (2): 425–430. Bibcode:2006PNAS..103..425G. doi:10.1073/pnas.0510013103. PMC 1324956
 . PMID 16407165. Retrieved 2007-10-16.
. PMID 16407165. Retrieved 2007-10-16.
- ↑ Smith, Hamilton O.; Clyde A. Hutchison; Cynthia Pfannkoch; J. Craig Venter (2003-12-23). "Generating a synthetic genome by whole genome assembly: {phi}X174 bacteriophage from synthetic oligonucleotides". Proceedings of the National Academy of Sciences. 100 (26): 15440–15445. Bibcode:2003PNAS..10015440S. doi:10.1073/pnas.2237126100. PMC 307586
 . PMID 14657399. Retrieved 2007-10-08.
. PMID 14657399. Retrieved 2007-10-08.
- 1 2 Gibson, B; Clyde A. Hutchison; Cynthia Pfannkoch; J. Craig Venter; et al. (2008-01-24). "Complete Chemical Synthesis, Assembly, and Cloning of a Mycoplasma genitalium Genome". Science. 319 (5867): 1215–20. Bibcode:2008Sci...319.1215G. doi:10.1126/science.1151721. PMID 18218864. Retrieved 2008-01-24.
- ↑ "Complete Genome of Synthetic Mycoplasma mycoides JCVI-syn1.0".
- 1 2 Lartigue C, Glass JI, Alperovich N, Pieper R, Parmar PP, Hutchison CA 3rd, Smith HO, Venter JC (Aug 3, 2007). "Genome transplantation in bacteria: changing one species to another". Science. 317 (5838): 632–8. Bibcode:2007Sci...317..632L. doi:10.1126/science.1144622. PMID 17600181.
- ↑ Avery OT, Macleod CM, McCarty M (Feb 1, 1944). "Studies on the Chemical Nature of the Substance Inducing Transformation of Pneumococcal Types : Induction of Transformation by a Desoxyribonucleic Acid Fraction Isolated From Pneumococcus Type III". J Exp Med. 79 (2): 137–58. doi:10.1084/jem.79.2.137. PMC 2135445
 . PMID 19871359.
. PMID 19871359. - 1 2 3 4 Gibson, D. G.; Glass, J. I.; Lartigue, C.; Noskov, V. N.; Chuang, R.-Y.; Algire, M. A.; Benders, G. A.; Montague, M. G.; Ma, L.; Moodie, M. M.; Merryman, C.; Vashee, S.; Krishnakumar, R.; Assad-Garcia, N.; Andrews-Pfannkoch, C.; Denisova, E. A.; Young, L.; Qi, Z.-Q.; Segall-Shapiro, T. H.; Calvey, C. H.; Parmar, P. P.; Hutchison, C. A.; Smith, H. O.; Venter, J. C. (20 May 2010). "Creation of a Bacterial Cell Controlled by a Chemically Synthesized Genome". Science. 329 (5987): 52–56. Bibcode:2010Sci...329...52G. doi:10.1126/science.1190719. PMID 20488990.
- ↑ Povolotskaya, IS; Kondrashov, FA (Jun 2010). "Sequence space and the ongoing expansion of the protein universe". Nature. 465 (7300): 922–6. doi:10.1038/nature09105.
- ↑ Blight KJ, Kolykhalov AA, Rice CM (Dec 8, 2000). "Efficient initiation of HCV RNA replication in cell culture". Science. 290 (5498): 1972–4. Bibcode:2000Sci...290.1972B. doi:10.1126/science.290.5498.1972. PMID 11110665.
- ↑ Cello, J; Paul, AV; Wimmer, E (Aug 2002). "Chemical synthesis of poliovirus cDNA: generation of infectious virus in the absence of natural template". Science. 297 (5583): 1016–8. doi:10.1126/science.1072266. PMID 12114528.
- ↑ Tawfik, Dan S (2010). "Messy biology and the origins of evolutionary innovations". Nature Chemical Biology. 6: 692–696. doi:10.1038/nchembio.441.
- ↑ US Patent Application: 20070122826
- ↑ Umenhoffer K, Fehér T, Balikó G, Ayaydin F, Pósfai J, Blattner FR, Pósfai G (2010). "Reduced evolvability of Escherichia coli MDS42, an IS-less cellular chassis for molecular and synthetic biology applications". Microb Cell Factories. 9: 38. doi:10.1186/1475-2859-9-38. PMC 2891674
 . PMID 20492662.
. PMID 20492662. - ↑ Pósfai G, Plunkett G 3rd, Fehér T, Frisch D, Keil GM, Umenhoffer K, Kolisnychenko V, Stahl B, Sharma SS, de Arruda M, Burland V, Harcum SW, Blattner FR (2006). "Emergent properties of reduced-genome Escherichia coli". Science. 312 (5776): 1044–6. Bibcode:2006Sci...312.1044P. doi:10.1126/science.1126439. PMID 16645050.
- ↑ Kolisnychenko V, Plunkett G 3rd, Herring CD, Fehér T, Pósfai J, Blattner FR, Pósfai G (April 2002). "Engineering a reduced Escherichia coli genome". Genome Res. 12 (4): 640–7. doi:10.1101/gr.217202. PMC 187512
 . PMID 11932248.
. PMID 11932248.
Popular press
- ↑ "Scientists create synthetic cell, version 1.0".
- 1 2 3 4 5 Pennisi E (May 2010). "Genomics. Synthetic genome brings new life to bacterium" (PDF). Science. 328 (5981): 958–9. doi:10.1126/science.328.5981.958. PMID 20488994.
- 1 2 3 4 Andrew Pollack, His Corporate Strategy: The Scientific Method, NYTimes, September 4, 2010
- 1 2 Pilkington, Ed (6 Oct 2009). "I am creating artificial life, declares US gene pioneer". London: The Guardian. Retrieved 23 Nov 2012.
- 1 2 Longest Piece of Synthetic DNA Yet, Scientific American News, 24 January 2008
- ↑ Wade, Nicholas (2007-06-29). "Scientists Transplant Genome of Bacteria". The New York Times. ISSN 0362-4331. Retrieved 2007-12-28.
- ↑ Henderson, Mark (May 21, 2010). "Scientists create artificial life in laboratory". The Times. London.
- ↑ Roberta Kwok (2010). "Genomics: DNA's master craftsmen". Nature. 468 (7320): 22–5. Bibcode:2010Natur.468...22K. doi:10.1038/468022a. PMID 21048740.
- ↑ "How scientists made 'artificial life'". BBC News. 2010-05-20. Retrieved 2010-05-21.
- ↑ Ken Shirriff (2010-06-10). "Using Arc to decode Venter's secret DNA watermark". Ken Shirriff's blog. Retrieved 2010-10-29.
- ↑ "Artificial life: Patent pending", The Economist, June 14, 2007. Retrieved October 7, 2007.
- ↑ Roger Highfield, "Man-made microbe 'to create endless biofuel'", Telegraph, June 8, 2007. Retrieved October 7, 2007.
- ↑ "First patent claimed on man-made life form, and challenged", World Science, June 7, 2007. Retrieved October 7, 2007.
- ↑ "Scarab Genomics LLC. Company web site.".
- ↑ Hutchison, Clyde A.; Chuang, Ray-Yuan; Noskov, Vladimir N.; Assad-Garcia, Nacyra; Deerinck, Thomas J.; Ellisman, Mark H.; Gill, John; Kannan, Krishna; Karas, Bogumil J. (2016-03-25). "Design and synthesis of a minimal bacterial genome". Science. 351 (6280): aad6253. doi:10.1126/science.aad6253. ISSN 0036-8075. PMID 27013737.
- ↑ First Minimal Synthetic Bacterial Cell. Astrobiology Web. March 24, 2016.
- 1 2 3 Yong, Ed (March 24, 2016). "The Mysterious Thing About a Marvelous New Synthetic Cell".
- ↑ Arcady R. Mushegian and Eugene V. Koonin (September 1996). "A minimal gene set for cellular life derived by comparison of complete bacterial genomes" (PDF). Proc. Natl. Acad. Sci. USA. 93: 10268–10273. doi:10.1073/pnas.93.19.10268. PMC 38373
 . PMID 8816789.
. PMID 8816789. - ↑ Zimmer, Carl. "And the Genomes Keep Shrinking...". National Geographic.