Total inorganic carbon
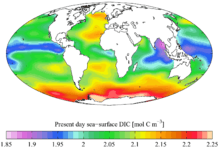
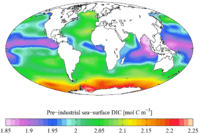
The total inorganic carbon (CT, or TIC) or dissolved inorganic carbon (DIC) is the sum of inorganic carbon species in a solution. The inorganic carbon species include carbon dioxide, carbonic acid, bicarbonate anion, and carbonate.[1] It is customary to express carbon dioxide and carbonic acid simultaneously as CO2* . CT is a key parameter when making measurements related to the pH of natural aqueous systems,[2] and carbon dioxide flux estimates.
- CT = [CO2*] + [HCO3−] + [CO32−]
where,
- CT is the total inorganic carbon
- [CO2*] is the sum of carbon dioxide and carbonic acid concentrations ( [CO2*] = [CO2] + [H2CO3])
- [HCO3−] is the bicarbonate concentration
- [CO32−] is the carbonate concentration
Each of these species are related by the following pH-driven chemical equilibria:
- CO2 + H2O ⇌ H2CO3 ⇌ H+ + HCO3− ⇌ 2H+ + CO32−
The concentrations of the different species of DIC (and which species is dominant) depends on the pH of the solution, as shown by a Bjerrum plot.
Total inorganic carbon is typically measured by the acidification of the sample which drives the equilibria to CO2. This gas is then sparged from solution and trapped, and the quantity trapped is then measured, usually by infrared spectroscopy.
See also
- Alkalinity (total alkalinity; AT)
- Bjerrum plot
- Fugacity (carbon dioxide fugacity; fCO2)
- Ocean acidification
- pH
- Total organic carbon
References
- ↑ C. Michael Hogan. 2010. Calcium. eds. A. Jorgensen, C. Cleveland. Encyclopedia of Earth. National Council for Science and the Environment.
- ↑ Stanley E. Manahan. 2005. Environmental chemistry. CRC Press