Lomustine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Gleostine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682207 |
| Pregnancy category |
|
| Routes of administration | Oral (capsules) |
| ATC code | L01AD02 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ~100% |
| Protein binding | 50% |
| Metabolism | Liver |
| Metabolites | Monoxydroxylated metabolites, trans-4-hydroxy-CCNU, cis-4-hydroxy-CCNU[1] |
| Biological half-life | 16–48 hours (metabolites) |
| Identifiers | |
| |
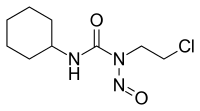
| Synonyms | 1-(2-chloroethyl)-3-cyclohexyl-1-nitrosourea |
| CAS Number |
13010-47-4 |
| PubChem (CID) | 3950 |
| IUPHAR/BPS | 7214 |
| DrugBank |
DB01206 |
| ChemSpider |
3813 |
| UNII |
7BRF0Z81KG |
| KEGG |
D00363 |
| ChEBI |
CHEBI:6520 |
| ChEMBL |
CHEMBL514 |
| ECHA InfoCard | 100.032.585 |
| Chemical and physical data | |
| Formula | C9H16ClN3O2 |
| Molar mass | 233.695 g/mol |
| 3D model (Jmol) | Interactive image |
| Melting point | 90 °C (194 °F) |
| |
| |
| | |
Lomustine (INN), abbreviated CCNU (original brand name (formerly available) is CeeNU, now marketed as Gleostine), is an alkylating nitrosourea compound used in chemotherapy. It is closely related to semustine and is in the same family as streptozotocin. It is a highly lipid-soluble drug[2] and thus crosses the blood-brain barrier. This property makes it ideal for treating brain tumors, which is its primary use. Lomustine has a long time to nadir (the time when white blood cells reach their lowest number).
Unlike carmustine, lomustine is administered orally. It is a monofunctional alkylating agent, alkylates both DNA and RNA, has the ability to cross-link DNA.[3] As with other nitrosoureas, it may also inhibit several key enzymatic processes by carbamoylation of amino acids in proteins.[4] Lomustine is cell-cycle nonspecific.
Rebranding
In 2014, the drug was re-launched and rebranded as Gleostine, manufactured by NextSource Biotechnology.[5][6]
References
- ↑ Lee, FY; Workman, P; Roberts, JT; Bleehen, NM (1985). "Clinical Pharmacokinetics of Oral CCNU (lomustine)". Cancer Chemotherapy and Pharmacology. 14 (2): 125–31. doi:10.1007/bf00434350. PMID 3971475.
- ↑ "BC Cancer Agency Cancer Drug Manual. Lomustine (CCNU; CeeNU)" (PDF). Retrieved 15 July 2016.
- ↑ Pizzo, PA; Poplack, DG, eds. (2006). Principles and Practice of Pediatric Oncology (5 ed.). Philadelphia: Lippincott Williams & Wilkins. p. 300. ISBN 9780781754927.
- ↑ "Gleostine (lomustine) Capsules, for Oral Use. Full Prescribing Information" (PDF). NextSource Biotechnology, LLC. Retrieved 15 July 2016.
- ↑ "NextSource Biotechnology Gains FDA Approval for Use of Tradename Gleostine (lomustine), an Anti-Cancer Chemotherapy Agent". www.prnewswire.com. NextSource Biotechnology. Retrieved 15 July 2016.
- ↑ "Gleostine (lomustine) Capsules — Healthcare Providers". NextSource Biotechnology. Retrieved 15 July 2016.
External links
- CeeNu (lomustine) Capsules data sheet published by the FDA
- Lomustine at the US National Library of Medicine Medical Subject Headings (MeSH)
- DDB 29525