Opisthorchis viverrini
| Opisthorchis viverrini | |
|---|---|
 | |
| An adult Opisthorchis viverrini prepared on a microscope slide | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Platyhelminthes |
| Class: | Trematoda |
| Order: | Opisthorchiida |
| Family: | Opisthorchiidae |
| Genus: | Opisthorchis |
| Species: | O. viverrini (Poirier, 1886) Stiles & Hassal, 1896 |
| Synonyms[1] | |
|
Distoma viverrini Poirier, 1886 | |
Opisthorchis viverrini, common name Southeast Asian liver fluke, is a trematode parasite from the family Opisthorchiidae that attacks the area of the bile duct. Infection is acquired when people ingest raw or undercooked fish.[2] Infection with the parasite is called opisthorchiasis. Opisthorchis viverrini infection also predisposes the infected for cholangiocarcinoma, a cancer of the gall bladder and/or its ducts.
Opisthorchis viverrini (together with Clonorchis sinensis and Opisthorchis felineus) is one of the three most medically important species in the family Opisthorchiidae.[3] In fact O. viverrini and C. sinensis are capable of causing cancer in humans, and are classified by the International Agency for Research on Cancer as a group 1 biological carcinogen in 2009.[4][5][6] O. viverrini is endemic throughout Thailand, the Lao People's Democratic Republic, Vietnam and Cambodia.[7] In Northern Thailand, it is widely distributed, with high prevalence in humans, while in Central Thailand there is low rate of prevalence.[8] The disease opisthorchiasis (caused by Opisthorchis viverrini) does not occur in southern Thailand.[8]
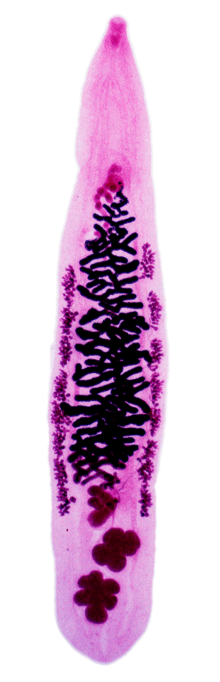
Description


The testes of an adult Opisthorchis viverrini are lobed[1] in comparison of dendritic testes of Clonorchis sinensis.[1]
The eggs of Opisthorchis viverrini are 30 × 12 μm in size[1] and they are slightly narrower and more regularly ovoid than in Clonorchis sinensis.[1] But eggs of Opisthorchis viverrini are visually indisgushiable in Kato technique smears from other eggs of flukes from other fluke family Heterophyidae.[9]
The metacercariae of Opisthorchis viverrini are brownish, elliptical with two nearly equal-sized suckers: the oral sucker and the ventral sucker.[9] They are 0.19–0.25 × 0.15–0.22 mm in size.[9]
Life cycle
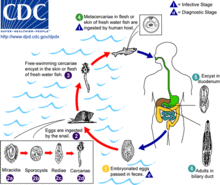
Opisthorchis viverrini is a hermaphroditic liver fluke.[7] Its life cycle is similar to the life cycle of Clonorchis sinensis.[7] It involves a freshwater snail, in which asexual reproduction takes place, and freshwater cyprinid fishes (family Cyprinidae) as intermediate hosts. Fish–eating (= piscivorous) mammals, including humans, dogs and cats, act as definitive hosts, in which sexual reproduction occurs.[7] As a result of poor sanitation practices and inadequate sewerage infrastructure, Opisthorchis viverrini-infected people pass the trematode's eggs in their feces into bodies of fresh water.[2]
First intermediate host

The first intermediate hosts include freshwater snails of the genus Bithynia.[10] The only known host is Bithynia siamensis (that include all its three subspecies).[1][11] Aquatic snails, which represent the first intermediate hosts of Opisthorchis viverrini, ingest the eggs from which the miracidia undergo asexual reproduction before a population of the free swimming larval stage, called a cercaria, is shed from the infected snails.[2]
Second intermediate host

The cercaria then locates a cyprinoid fish, encysts in the fins, skin and musculature of the fish, and becomes a metacercaria.[2] Habitats of second intermediate hosts of Opisthorchis viverrini include freshwater habitats with stagnant or slow-moving waters (ponds, river, aquaculture, swamps, rice fields).[12]
In 1965 there were known 9 fish hosts of Opisthorchis viverrini.[13] Up to 2002 there were known 15 species of fishes from 7 genera of the family Cyprinidae, that serves as second intermediate host.[1] Further research by Rim et al. (2008)[9] showed additional five more host species:
- Puntius brevis[9]
- Puntius gonionotus – synonym: Barbonymus gonionotus, Java barb[9]
- Puntius orphoides[1][9]
- Puntius proctozysron – synonym: Puntioplites proctozystron[9]
- Puntius viehoeveri – synonym: Barbonymus gonionotus[9]
- Hampala dispar[1][9]
- Hampala macrolepidota[9]
- Cyclocheilichthys armatus[9] – synonym: Cyclocheilichthys siaja[14]
- Cyclocheilichthys repasson[9]
- Labiobarbus lineatus[9]
- Esomus metallicus[9]
- Mystacoleucus marginatus[9]
- Puntioplites falcifer[9]
- Onychostoma elongatum[9]
- Osteochilus hasseltii[9]
- Hypsibarbus lagleri[9]
- Barbodes gonionotus – synonym: Barbonymus gonionotus[9]
Definitive host
The metacercarial stage is infective to humans and other fish-eating mammals[2] including dogs, cats,[7] rats, and pigs.[16] The natural definitive host is the leopard cat (Prionailurus bengalensis).[1] The young adult worm escapes from the metacercarial cyst in the upper small intestine and then migrates through the ampulla of Vater into the biliary tree, where it develops to sexual maturity over four to six weeks, thus completing the life cycle.[2]
Fish contain more metacercaria from September to February, before the dry season[1][13] and this is the period, when humans are usually infected.[1] Infection is acquired when people ingest raw or undercooked fish.[2] Dishes of raw fish are common in the cuisine of Laos and the cuisine of Thailand: koi-pla, raw fish in spicy salad larb-pla,[11] salted semi-fermented fish dishes called pla-ra (pla ra),[1] pla som[11] and Som fak.[9]
The adult worms, which are hermaphrodites, can live for many years in the liver, even decades, shedding as many as 200 eggs per day which pass out via bile into the chyme and feces.[2] The lifespan of Opisthorchis viverrini is over 10 years.[14]
Opisthorchis viverrini secretes a granulin-like growth protein especially in its gut and integument.[17]
Effect on human health
Medical care and loss of wages caused by Opisthorchis viverrini in Laos and in Thailand costs about $120 million annually[1] or $120 million per year can cost Northeast Thailand only.[3]
Infections with Opisthorchis viverrini and of other liver flukes in Asia affect the poor and poorest people.[18] Opisthorchiasis has received less attention in comparison to other diseases, and it is a neglected disease in Asia.[18]
Genetics
Currently, a total of only ~5,000[2] expressed sequence tags (ESTs) are publicly available for Opisthorchis viverrini, a dataset far too small to give sufficient insights into transcriptomes for the purpose of supporting genomic and other fundamental molecular research.[7]
Although the genome size of Opisthorchis viverrini has not yet been reported, it is known to have six pairs of chromosomes, i.e. 2n = 12.[2]
References
This article incorporates CC-BY-2.5 text from references [7][15][19] and CC-BY-2.0 text from the reference.[2]
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Muller R. & Wakelin D. (2002). Worms and human disease. CABI. pp. 43–44.
- 1 2 3 4 5 6 7 8 9 10 11 Laha, T.; Pinlaor, P.; Mulvenna, J.; Sripa, B.; Sripa, M.; Smout, M. J.; Gasser, R. B.; Brindley, P. J.; Loukas, A. (2007). "Gene discovery for the carcinogenic human liver fluke, Opisthorchis viverrini". BMC Genomics. 8: 189. doi:10.1186/1471-2164-8-189. PMC 1913519
 . PMID 17587442..
. PMID 17587442.. - 1 2 King, S.; Scholz, T. Š. (2001). "Trematodes of the family Opisthorchiidae: A minireview". The Korean Journal of Parasitology. 39 (3): 209–221. doi:10.3347/kjp.2001.39.3.209. PMC 2721069
 . PMID 11590910.
. PMID 11590910. - ↑ Kaewpitoon N, Kaewpitoon SJ, Pengsaa P, Sripa B (2008). "Opisthorchis viverrini: the carcinogenic human liver fluke". World J Gastroenterol. 14 (5): 666–674. doi:10.3748/wjg.14.666. PMC 2683991
 . PMID 18205254.
. PMID 18205254. - ↑ Sripa B, Brindley PJ, Mulvenna J, Laha T, Smout MJ, Mairiang E, Bethony JM, Loukas A (2012). "The tumorigenic liver fluke Opisthorchis viverrini--multiple pathways to cancer". Trends in Parasitology. 28 (10): 395–407. doi:10.1016/j.pt.2012.07.006. PMC 3682777
 . PMID 22947297.
. PMID 22947297. - ↑ American Cancer Society (2013). "Known and Probable Human Carcinogens". cancer.org. American Cancer Society, Inc. Retrieved 2013-06-02.
- 1 2 3 4 5 6 7 Young, N. D.; Campbell, B. E.; Hall, R. S.; Jex, A. R.; Cantacessi, C.; Laha, T.; Sohn, W. M.; Sripa, B.; Loukas, A.; Brindley, P. J.; Gasser, R. B. (2010). Jones, Malcolm K, ed. "Unlocking the Transcriptomes of Two Carcinogenic Parasites, Clonorchis sinensis and Opisthorchis viverrini". PLoS Neglected Tropical Diseases. 4 (6): e719. doi:10.1371/journal.pntd.0000719. PMC 2889816
 . PMID 20582164.
. PMID 20582164. - 1 2 Jongsuksuntigul, P.; Imsomboon, T. (2003). "Opisthorchiasis control in Thailand". Acta Tropica. 88 (3): 229–232. doi:10.1016/j.actatropica.2003.01.002. PMID 14611877.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 Rim, H. J.; Sohn, W. M.; Yong, T. S.; Eom, K. S.; Chai, J. Y.; Min, D. Y.; Lee, S. H.; Hoang, E. H.; Phommasack, B.; Insisengmay, S. (2008). "Fishborne Trematode Metacercariae Detected in Freshwater Fish from Vientiane Municipality and Savannakhet Province, Lao PDR". The Korean Journal of Parasitology. 46 (4): 253–260. doi:10.3347/kjp.2008.46.4.253. PMC 2612611
 . PMID 19127332..
. PMID 19127332.. - ↑ Tohamy A. A.; Mohamed S. M. (2006). "Chromosomal studies on two Egyptian freshwater snails, Cleopatra and Bithynia (Mollusca-Prosobranchiata)" (PDF). Arab J. Biotech. 9 (1): 17–26.
- 1 2 3 World Health Organization (1995). Control of Foodborne Trematode Infection. WHO Technical Report Series. 849. PDF part 1, PDF part 2. page 89-91.
- ↑ Keiser, J.; Utzinger, J. R. (2007). "Artemisinins and synthetic trioxolanes in the treatment of helminth infections". Current Opinion in Infectious Diseases. 20 (6): 605–612. doi:10.1097/QCO.0b013e3282f19ec4. PMID 17975411..
- 1 2 Wykoff, D. E.; Harinasuta, C.; Juttijudata, P.; Winn, M. M. (1965). "Opisthorchis Viverrini in Thailand--The Life Cycle and Comparison with O. Felineus". The Journal of Parasitology. 51 (2): 207–214. doi:10.2307/3276083. JSTOR 3276083. PMID 14275209., JSTOR.
- 1 2 Harinasuta, C.; Harinasuta, T. (1984). "Opisthorchis viverrini: Life cycle, intermediate hosts, transmission to man and geographical distribution in Thailand". Arzneimittel-Forschung. 34 (9B): 1164–1167. PMID 6542383.
- 1 2 Sripa, B.; Kaewkes, S.; Sithithaworn, P.; Mairiang, E.; Laha, T.; Smout, M.; Pairojkul, C.; Bhudhisawasdi, V.; Tesana, S.; Thinkamrop, B.; Bethony, J. M.; Loukas, A.; Brindley, P. J. (2007). "Liver Fluke Induces Cholangiocarcinoma". PLoS Medicine. 4 (7): e201. doi:10.1371/journal.pmed.0040201. PMC 1913093
 . PMID 17622191..
. PMID 17622191.. - ↑ Chai J. Y.; Darwin Murrell K.; Lymbery A. J. (2005). "Fish-borne parasitic zoonoses: Status and issues". International Journal for Parasitology. 35 (11-12): 1233–1254. doi:10.1016/j.ijpara.2005.07.013.
- ↑ Smout, M. J.; Laha, T.; Mulvenna, J.; Sripa, B.; Suttiprapa, S.; Jones, A.; Brindley, P. J.; Loukas, A. (2009). Pearce, Edward J, ed. "A Granulin-Like Growth Factor Secreted by the Carcinogenic Liver Fluke, Opisthorchis viverrini, Promotes Proliferation of Host Cells". PLoS Pathogens. 5 (10): e1000611. doi:10.1371/journal.ppat.1000611. PMC 2749447
 . PMID 19816559..
. PMID 19816559.. - 1 2 Sripa, B. (2008). Loukas, Alex, ed. "Concerted Action is Needed to Tackle Liver Fluke Infections in Asia". PLoS Neglected Tropical Diseases. 2 (5): e232. doi:10.1371/journal.pntd.0000232. PMC 2386259
 . PMID 18509525..
. PMID 18509525.. - ↑ Traub, R. J.; MacAranas, J.; Mungthin, M.; Leelayoova, S.; Cribb, T.; Murrell, K. D.; Thompson, R. C. A. (2009). Sripa, Banchob, ed. "A New PCR-Based Approach Indicates the Range of Clonorchis sinensis Now Extends to Central Thailand". PLoS Neglected Tropical Diseases. 3 (1): e367. doi:10.1371/journal.pntd.0000367. PMC 2614470
 . PMID 19156191..
. PMID 19156191..
External links
| Wikimedia Commons has media related to Opisthorchis viverrini. |
- Public Health Agency of Canada Opisthorchis spp. - Pathogen Safety Data Sheet http://www.phac-aspc.gc.ca/lab-bio/res/psds-ftss/msds115e-eng.php
- "Opisthorchis viverrini". National Center for Biotechnology Information (NCBI).
- "Opisthorchiasis — Opisthorchis felineus Opisthorchis viverrini". DPDx Laboratory Identification of Parasites of Public Health Concern. Centers for Disease Control and Prevention.
- "Introduction — Opisthorchiasis". Parasites and Pestilence. The Program in Human Biology, Stanford University.
- Riganti, M.; Pungpak, S.; Punpoowong, B.; Bunnag, D.; Harinasuta, T. (1989). "Human pathology of Opisthorchis viverrini infection: A comparison of adults and children". The Southeast Asian journal of tropical medicine and public health. 20 (1): 95–100. PMID 2772709.
- Upatham, E. S.; Viyanant, V. (2003). "Opisthorchis viverrini and opisthorchiasis: A historical review and future perspective". Acta Tropica. 88 (3): 171–176. doi:10.1016/j.actatropica.2003.01.001. PMID 14611871.
- Andrews, R. H.; Sithithaworn, P.; Petney, T. N. (2008). "Opisthorchis viverrini: An underestimated parasite in world health". Trends in Parasitology. 24 (11): 497–501. doi:10.1016/j.pt.2008.08.011. PMC 2635548
 . PMID 18930439.
. PMID 18930439. - Kaewpitoon, N.; Kaewpitoon, S. J.; Pengsaa, P.; Sripa, B. (2008). "Opisthorchis viverrini: The carcinogenic human liver fluke". World Journal of Gastroenterology. 14 (5): 666–674. doi:10.3748/wjg.14.666. PMC 2683991
 . PMID 18205254.
. PMID 18205254. - Kaewkes, S. (2003). "Taxonomy and biology of liver flukes". Acta tropica. 88 (3): 177–186. doi:10.1016/j.actatropica.2003.05.001. PMID 14611872.
- Adam, R.; Arnold, H.; Hinz, E.; Storch, V. (1995). "Morphology and ultrastructure of the redia and pre-emergent cercaria of Opisthorchis viverrini (Trematoda: Digenea) in the intermediate host Bithynia siamensis goniomphalus (Prosobranchia: Bithyniidae)". Applied Parasitology. 36 (2): 136–154. PMID 7550441.
- Inatomi, S.; Tongu, Y.; Sakumoto, D.; Suguri, S.; Itano, K. (1971). "The ultrastructure of helminth. VI. The body wall of Opisthorchis viverrini (Poirier, 1886)". Acta medicinae Okayama. 25 (2): 129–142. PMID 4333630..
