Biphenylindanone A
Biphenylindanone A
 |
| Clinical data |
|---|
| ATC code |
none |
|---|
| Identifiers |
|---|
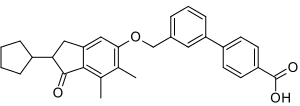
- 3’-(((2-cyclopentyl-6,7-dimethyl-1-oxo-2,3-dihydro- 1H-inden-5-yl)oxy)methyl)biphenyl-4-carboxylic acid
|
| PubChem (CID) |
9868580 |
|---|
| IUPHAR/BPS |
3954 |
|---|
| ChemSpider |
8044271  Y Y |
|---|
| ChEMBL |
CHEMBL593013  Y Y |
|---|
| Chemical and physical data |
|---|
| Formula |
C30H30O4 |
|---|
| Molar mass |
454.556 g/mol |
|---|
| 3D model (Jmol) |
Interactive image |
|---|
c3cc(C(O)=O)ccc3-c(ccc4)cc4COc(c1C)cc(c(C2=O)c1C)CC2C5CCCC5
|
InChI=1S/C30H30O4/c1-18-19(2)28-25(15-26(29(28)31)22-7-3-4-8-22)16-27(18)34-17-20-6-5-9-24(14-20)21-10-12-23(13-11-21)30(32)33/h5-6,9-14,16,22,26H,3-4,7-8,15,17H2,1-2H3,(H,32,33)  Y YKey:KMKBEESNZAPKMP-UHFFFAOYSA-N  Y Y
|
| (verify) |
|---|
Biphenylindanone A (BINA, LS-193,571) is a research agent which acts as a potent and selective positive allosteric modulator for the group II metabotropic glutamate receptor subtype mGluR2.
In animal studies it showed anxiolytic and antipsychotic effects,[1] and blocked the effects produced by the hallucinogenic drug DOB. BINA and other selective mGluR2 positive modulators have therefore been suggested as a novel class of drugs for the treatment of schizophrenia which may have superior properties to traditional antipsychotic drugs.[2]
BINA decreases cocaine self-administration in rats, with no effect on food self-administration, and is in regard to this discrimination superior to the mGluR2/3 agonist LY-379,268[3]
References
- ↑ Galici R; Jones CK; Hemstapat K; et al. (2006). "Biphenyl-indanone A, a positive allosteric modulator of the metabotropic glutamate receptor subtype 2, has antipsychotic- and anxiolytic-like effects in mice". The Journal of Pharmacology and Experimental Therapeutics. 318 (1): 173–85. doi:10.1124/jpet.106.102046. PMID 16608916.
- ↑ Benneyworth MA, Xiang Z, Smith RL, Garcia EE, Conn PJ, Sanders-Bush E (2007). "A selective positive allosteric modulator of metabotropic glutamate receptor subtype 2 blocks a hallucinogenic drug model of psychosis". Molecular Pharmacology. 72 (2): 477–84. doi:10.1124/mol.107.035170. PMID 17526600.
- ↑ Jin, Xinchun; Semenova, Svetlana; Yang, Li; Ardecky, Robert; Sheffler, Douglas J; Dahl, Russell; Conn, P Jeffrey; Cosford, Nicholas DP; Markou, Athina (2010). "The mGluR2 Positive Allosteric Modulator BINA Decreases Cocaine Self-Administration and Cue-Induced Cocaine-Seeking and Counteracts Cocaine-Induced Enhancement of Brain Reward Function in Rats". Neuropsychopharmacology. 35 (10): 2021–36. doi:10.1038/npp.2010.82. PMC 2922422
 . PMID 20555310.
. PMID 20555310.
|
|---|
|
Receptor
(ligands) | | AMPA | |
|---|
| | NMDA |
- Antagonists: Competitive antagonists: AP5 (APV)
- AP7
- CGP-37849
- CGP-39551
- CGP-39653
- CGP-40116
- CGS-19755
- CPP
- LY-233,053
- LY-235,959
- LY-274,614
- MDL-100,453
- Midafotel (d-CPPene)
- NPC-12,626
- NPC-17,742
- PBPD
- PEAQX
- Perzinfotel
- PPDA
- SDZ-220581
- Selfotel; Noncompetitive antagonists: ARR-15,896
- Caroverine
- Dexanabinol
- FPL-12495
- FR-115,427
- Hodgkinsine
- Magnesium
- MDL-27,266
- NPS-1506
- Psychotridine
- Zinc; Uncompetitive pore blockers: 2-MDP
- 3-HO-PCP
- 3-MeO-PCE
- 3-MeO-PCMo
- 3-MeO-PCP
- 4-MeO-PCP
- 8A-PDHQ
- 18-MC
- α-Endopsychosin
- Alaproclate
- Amantadine
- Aptiganel
- Arketamine
- ARL-12,495
- ARL-15,896-AR
- ARL-16,247
- Budipine
- Conaridine
- Delucemine
- Dexoxadrol
- Dextrallorphan
- Dieticyclidine
- Diphenidine
- Dizocilpine
- Ephenidine
- Esketamine
- Etoxadrol
- Eticyclidine
- Fluorolintane
- Gacyclidine
- Ibogaine
- Ibogamine
- Indantadol
- Ketamine
- Ketobemidone
- Lanicemine
- Loperamide
- Memantine
- Methadone (Levomethadone)
- Methorphan (Dextromethorphan
- Levomethorphan)
- Methoxetamine
- Methoxphenidine
- Milnacipran
- Morphanol (Dextrorphan
- Levorphanol)
- NEFA
- Neramexane
- Nitromemantine
- Nitrous oxide
- Noribogaine
- Norketamine
- Orphenadrine
- PCPr
- Pethidine (meperidine)
- Phencyclamine
- Phencyclidine
- Propoxyphene
- Remacemide
- Rhynchophylline
- Rimantadine
- Rolicyclidine
- Sabeluzole
- Tabernanthine
- Tenocyclidine
- Tiletamine
- Tramadol
- Xenon; Glycine site antagonists: 4-Cl-KYN (AV-101)
- 5,7-DCKA
- 7-CKA
- ACC
- ACEA-1011
- ACEA-1328
- AV-101
- Carisoprodol
- CGP-39653
- CNQX
- DNQX
- Felbamate
- Gavestinel
- GV-196,771
- Kynurenic acid
- Kynurenine
- L-689,560
- L-701,324
- Licostinel (ACEA-1021)
- LU-73,068
- MDL-105,519
- Meprobamate
- MRZ 2/576
- PNQX
- ZD-9379; NR2B subunit antagonists: Besonprodil
- CERC-301 (MK-0657)
- CO-101,244 (PD-174,494)
- Eliprodil
- Haloperidol
- Ifenprodil
- Isoxsuprine
- Nylidrin
- Ro8-4304
- Ro25-6981
- Traxoprodil; Polyamine site antagonists: Arcaine
- Co 101676
- Diaminopropane
- Diethylenetriamine
- Huperzine A
- Putrescine
- Ro 25-6981; Unclassified/unsorted antagonists: Bumetanide
- Chloroform
- Cyclopropane
- D-αAA
- Diethyl ether
- Enflurane
- Ethanol
- Flufenamic acid
- Flupirtine
- Furosemide
- Halothane
- Isoflurane
- Metaphit
- Methoxyflurane
- Niflumic acid
- Pentamidine isethionate
- Piretanide
- Toluene
- Transcrocetin (saffron)
- Trichloroethane
- Trichloroethanol
- Trichloroethylene
- Xylene
|
|---|
| | Kainate | |
|---|
| | mGlu1 | |
|---|
| | mGlu2 | |
|---|
| | mGlu3 | |
|---|
| | mGlu4 |
- Antagonists: CPPG
- MAP4
- MPPG
- MSOP
- MTPG
- UBP-1112
|
|---|
| | mGlu5 | |
|---|
| | mGlu6 |
- Antagonists: CPPG
- MAP4
- MPPG
- MSOP
- MTPG
- UBP-1112
|
|---|
| | mGlu7 |
- Antagonists: CPPG
- MAP4
- MMPIP
- MPPG
- MSOP
- MTPG
- UBP-1112
|
|---|
| | mGlu8 |
- Antagonists: CPPG
- MAP4
- MPPG
- MSOP
- MTPG
- UBP-1112
|
|---|
|
|---|
|
Transporter
(blockers) | |
|---|
|
Enzyme
(inhibitors) | |
|---|
|
| Others | |
|---|
|
See also: GABAergics • GHBergics • Glycinergics |

 . PMID 20555310.
. PMID 20555310.